Report to the Subcommittee on Border Management, Federal Workforce, and Regulatory Affairs, Committee on Homeland Security and Governmental Affairs, U.S. Senate
United States Government Accountability Office
A report to the Subcommittee on Border Management, Federal Workforce, and Regulatory Affairs, Committee on Homeland Security and Governmental Affairs, U.S. Senate
For more information, contact: Yvonne D. Jones at JonesY@gao.gov.
What GAO Found
For about a quarter of the major rules issued from January 21, 2021, to January 20, 2025 (119 of 462 rules), agencies published effective dates inconsistent with the Congressional Review Act’s (CRA) 60-day waiting period. The waiting period begins when the Federal Register has published the rule or the House and the Senate have received paper copies, whichever is later, and does not apply under certain exceptions, such as when agencies claim good cause.
The CRA requires rules to be received by Congress but does not further define what constitutes receipt by Congress or establish what must occur for the House and the Senate to receive a rule. Officials from all four agencies described challenges with understanding what counts as the date Congress receives copies of rules. Amending the CRA to be more precise about the date of receipt, such as by specifying that receipt occurs when agencies deliver rules to an identified office or location, would help allay the confusion agencies face. Doing so would promote the CRA’s goal of ensuring a specific minimum delay in the effective dates of major rules.
Department of Energy (DOE) officials told GAO that they set effective dates for some major rules at 75 days from the date of publication in the Federal Register, instead of 60 days, which allows Congress additional time to process and receive the rules. However, no department policy formalizes this practice. GAO found that both DOE and the Department of the Treasury could have increased the number of major rules with effective dates consistent with the CRA by routinely allowing this additional time.
The Department of Health and Human Services (HHS), unlike the other selected agencies, sometimes uses a different date to start the clock on the 60-day delay period. HHS officials told GAO that for some rules, they set the effective date 60 days from the date the Federal Register displays a version of the rule for public inspection, instead of using the date of official publication, as the CRA requires. HHS officials stated they did so because these rules are long and complex and can take weeks to process for publication. As a result, Congress had less time to consider these rules before they were put into effect.
Why GAO Did This Study
Under the CRA, rules that are likely to have large economic impacts (major rules) are subject to a 60-day waiting period before they can take effect. In previous reports, GAO found that agencies frequently set effective dates (i.e., the dates when rules are to take effect) earlier than allowed by the CRA.
GAO was asked to examine agencies’ implementation of the CRA’s 60-day delay for major rules. This report examines, among other issues, (1) the number of major rules issued from January 21, 2021, to January 20, 2025, with stated effective dates inconsistent with the CRA; and (2) challenges selected agencies experienced in setting dates consistent with the CRA.
GAO identified rules with stated effective dates inconsistent with the CRA using its major rule reports, the Federal Register, and the Congressional Record. GAO selected four agencies with the largest number of rules that were inconsistent with the CRA—the Department of Agriculture, DOE, HHS, and Treasury. GAO reviewed documentation and interviewed agency officials on their policies and challenges they may have experienced.
What GAO Recommends
GAO recommends that Congress consider amending the CRA to specify when rules count as received. GAO also recommends that Treasury and DOE amend their policies to increase the time between the Federal Register publication date and the stated effective date of major rules and that HHS take steps to ensure its published effective dates for major rules are consistent with the CRA. DOE did not provide comments. Treasury and HHS agreed that congressional action is needed and stated they intend to take actions consistent with our recommendations.
|
Abbreviations |
|
|
CFR |
Code of Federal Regulations |
|
CRA |
Congressional Review Act |
|
DOE |
Department of Energy |
|
EPA |
Environmental Protection Agency |
|
HHS |
Department of Health and Human Services |
|
IRS |
Internal Revenue Service |
|
OFR |
Office of the Federal Register |
|
SEC |
Securities and Exchange Commission |
|
USDA |
Department of Agriculture |
This is a work of the U.S. government and is not subject to copyright protection in the United States. The published product may be reproduced and distributed in its entirety without further permission from GAO. However, because this work may contain copyrighted images or other material, permission from the copyright holder may be necessary if you wish to reproduce this material separately.
April 16, 2026
The Honorable James Lankford
Chairman
The Honorable John Fetterman
Ranking Member
Subcommittee on Border Management, Federal Workforce, and Regulatory Affairs
Committee on Homeland Security and Governmental Affairs
United States Senate
Federal agencies issue thousands of rules each year. Some have local and temporary effects, such as a Coast Guard rule establishing a safety zone in a river for a New Year’s Eve fireworks show.[1] Others may affect millions of Americans, such as Federal Communications Commission rules on routing of 911 calls and blocking of unwanted text messages.[2]
Under the Congressional Review Act (CRA), rules that are likely to have large economic impacts (i.e., major rules) are subject to a 60-day waiting period before they can take effect.[3] According to some of the Members of Congress who sponsored these provisions, the waiting period is designed to provide Congress with an opportunity to disapprove these rules before regulated parties must invest the significant resources necessary to comply with them.[4] The CRA also created special procedures for Congress to pass joint resolutions disapproving rules issued by agencies.[5] Since the CRA was enacted in 1996, hundreds of resolutions of disapproval have been introduced, and 42 rules have been overturned by such resolutions.[6] A joint resolution of disapproval, when signed by the President, nullifies the rule, whether or not the rule has already taken effect.
The CRA includes exceptions to the 60-day delay requirement. One of these exceptions allows an agency to make rules effective without delay or with a delay of less than 60 days when the agency finds for “good cause” (and incorporates the finding and a brief statement of reasons therefor in the rule issued) that notice and public procedure on the rule are impracticable, unnecessary, or contrary to the public interest.[7]
In previous reports, we found that agencies frequently published rules’ effective dates (i.e., the dates when rules are to take effect) earlier than allowed by the CRA. Specifically, we reported that, with some year-to-year variation, this inconsistency between the CRA and the effective dates of rules persisted during transitions between presidential administrations and in selected nontransition periods in the Clinton, Bush, Obama, and first Trump administrations, from September 1996 to January 2021.[8]
You asked us to examine agencies’ implementation of the CRA’s 60-day delay for major rules. This report examines (1) the number of major rules issued from January 21, 2021, to January 20, 2025, that had effective dates inconsistent with the CRA; (2) the distribution by agency and by year of rules with inconsistent effective dates; and (3) challenges selected agencies experienced in meeting CRA requirements for the effective dates of major rules.[9]
To address our objectives, we reviewed the major rule report GAO prepared for Congress for each of the 462 rules within the 4-year scope of our review, which represented the full term of the Biden administration.[10] The CRA requires GAO to issue a major rule report within 15 days regarding each major rule submitted by an agency.[11] For certain rules, we also reviewed the publications in the Federal Register and Congressional Record to obtain information about key dates that may not have been available at the time the major rule report was issued. We did not evaluate the agencies’ decisions regarding procedural requirements or their determinations regarding the potential effects of their regulations—including determinations of which rules were major.
We identified rules that claimed a statutory exception to the 60-day delay requirement using the summary information provided in the major rule reports. For rules that agencies claimed were covered by the CRA’s good cause exception, we examined the reasons agencies provided when claiming this exception. For this analysis, we placed the agencies’ provided reasons into categories we developed based on our previous work on rulemaking. We added additional categories as needed. For example, we included a category for good cause claims related to the COVID-19 pandemic.
To identify challenges that selected agencies experienced in meeting CRA requirements, we selected the four agencies with the greatest number of rules that had stated effective dates inconsistent with the CRA’s 60-day delay requirement based on data as of October 2024, the most recent data available at the time we initiated our review. These agencies were the Departments of Agriculture (USDA), Energy (DOE), Health and Human Services (HHS), and the Treasury. We obtained and reviewed documentation and interviewed officials from these agencies regarding their policies and procedures for submitting rules to Congress, GAO, and the Office of the Federal Register (OFR) and challenges they may have experienced.
In addition, we interviewed the House and Senate Parliamentarians and their staff regarding procedures for receiving rules submitted by agencies. We also reviewed documents and interviewed officials from the Office of Management and Budget and OFR, which is part of the National Archives and Records Administration. In addition, we interviewed knowledgeable individuals from academic institutions, the Administrative Conference of the United States, and the Congressional Research Service.[12] More information on our objectives, scope, and methodology is provided in appendix I.
We conducted this performance audit from September 2024 to April 2026 in accordance with generally accepted government auditing standards. Those standards require that we plan and perform the audit to obtain sufficient, appropriate evidence to provide a reasonable basis for our findings and conclusions based on our audit objectives. We believe that the evidence obtained provides a reasonable basis for our findings and conclusions based on our audit objectives.
Background
The CRA was enacted in 1996 in part to better ensure that Congress has an opportunity to review rules before they take effect and to disapprove any rule to which Congress objects. The act established expedited procedures by which Congress may disapprove agencies’ rules by introducing a resolution of disapproval that, if adopted by both Houses of Congress and signed by the President, can nullify an agency’s action.[13]
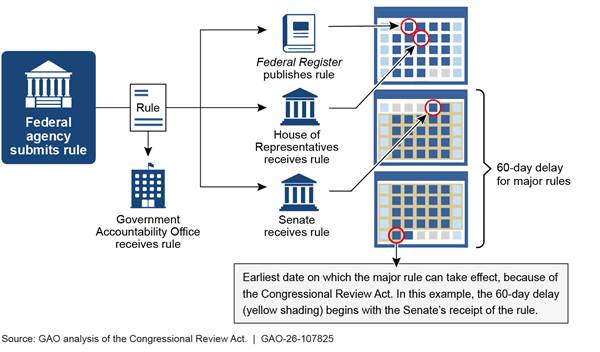
The CRA also established requirements for agencies. For each rule they issue, agencies must submit to GAO and to each House of Congress a report containing a copy of the rule, a concise general statement relating to the rule, including whether it is a major rule, and the rule’s proposed effective date. They must also provide to GAO and make available to each House of Congress a complete copy of the rule’s cost-benefit analysis, agency actions relevant to certain statutory requirements for rulemaking, and any other relevant information or requirements under other acts or executive orders.[14] GAO accepts these submissions electronically by email, while the House of Representatives and the Senate require paper copies.
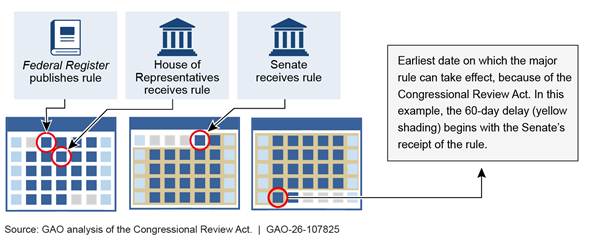
Major rules are subject to additional requirements. The CRA establishes a 60-day delay in the rule’s effective date for major rules.[15] The delay begins on the date the rule is received by Congress or the date the rule is published in the Federal Register, whichever is later (see fig. 1).[16]
Note: This is an example of a major rule that is published in the Federal Register and is not covered by an exception from the 60-day delay requirement. For illustrative purposes, it shows receipt by the Senate as occurring after receipt by the House and publication in the Federal Register. These events can occur in any order, and the 60-day delay begins from the latest of them.
There are two exceptions within the CRA to the 60-day delay requirement for major rules. First, as discussed above, the delay does not apply if the agency finds for good cause that notice and public procedure on the rule are impracticable, unnecessary, or contrary to the public interest. The agency must incorporate a statement of the findings and its reasons in the rule issued. For example, agencies have invoked this good cause exception for rules that they claimed needed to be issued quickly because of a statutory deadline or an emergency. Second, the 60-day delay does not apply to certain rules related to hunting, fishing, or camping.[17]
The CRA also requires GAO to provide Congress with a report on each major rule. These reports include an assessment of the issuing agency’s compliance with the procedural steps required by various laws and executive orders governing the rulemaking process.[18]
The Congressional Record is a record of Congress’s proceedings and debates.[19] The receipt of rules submitted by agencies pursuant to the CRA is recorded in the Executive Communications section of the Congressional Record, with separate entries for the House and the Senate.
The Federal Register is the federal government’s official daily publication for rules, including proposed and final rules; federal agency notices; and presidential documents. Rules published in the Federal Register include effective dates provided by the agency issuing the rule (see fig. 2). For rules that affect the content of the Code of Federal Regulations (CFR), the CFR is generally updated on the stated effective date provided by the agency.
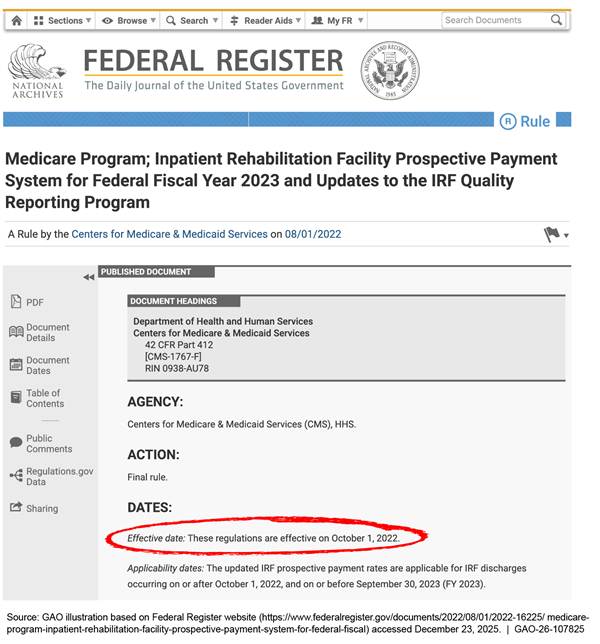
Note: For this example, we modified some elements of the web page due to space limitations.
OFR displays documents, including rules, for the public to review before they are printed in the Federal Register; this is known as public inspection. OFR generally makes rules available for public inspection at least 1 business day before they are officially published. Rules currently posted for public inspection can be viewed on the Federal Register website, https://www.federalregister.gov (see fig. 3).
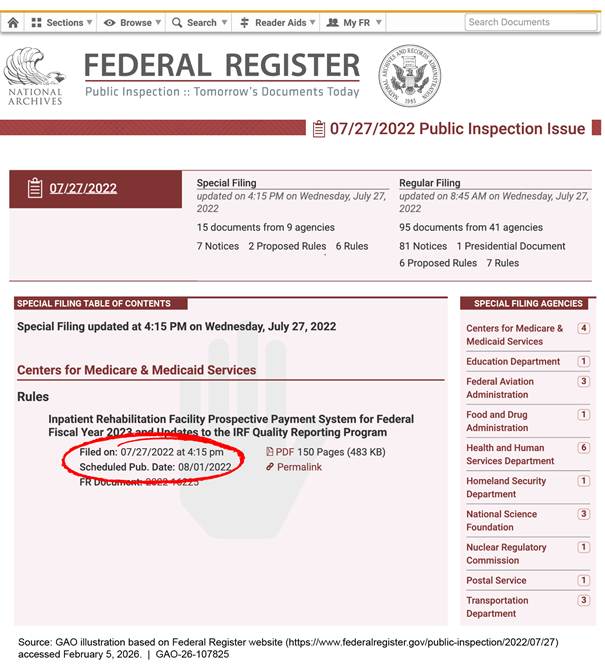
Note: For this example, we modified some elements of the web page due to space limitations.
Published Effective Dates Were Inconsistent with the CRA for About a Quarter of Major Rules in Our Review Period
For about a quarter of the rules issued during the Biden administration (January 21, 2021, to January 20, 2025), agencies provided less than the required 60-day delay before the stated effective date of the rule without claiming an exception.[20] Our analysis showed that of the total 462 major rules that agencies issued and submitted to GAO between January 21, 2021, and January 20, 2025, 119 (about 26 percent) had stated effective dates that were inconsistent with the CRA. Specifically, each of the 119 rules provided less than 60 days of delay and did not include findings related to either of the CRA’s two exceptions.
The magnitude of the delays in the rules’ effective dates varied. For example, of the 119 major rules with effective dates that were inconsistent with the CRA, 19 provided between 56 and 59 days of delay, thereby missing the standard 60-day time frame by less than 5 days. Another 57 rules provided between 30 and 55 days of delay, thereby missing the standard 60-day time frame by 5 to 30 days. The remaining 43 rules provided less than 30 days of delay, missing the standard time frame by more than 30 days, and in some of these cases, Congress did not receive the rules until after the stated effective date of the rule.
We also found that, for 82 of the total 462 major rules agencies submitted (about 18 percent), agencies claimed an exception from the 60-day delay requirement. Specifically, for 70 of the 82 rules, agencies claimed the CRA’s good cause exception, which requires a finding that notice and public procedure are impracticable, unnecessary, or contrary to the public interest.[21] Another 12 rules claimed the exception for certain rules related to hunting, fishing, or camping (see fig. 4).
Figure 4: Consistency with Congressional Review Act 60-Day Delay Requirement for Major Rules Issued January 21, 2021, through January 20, 2025
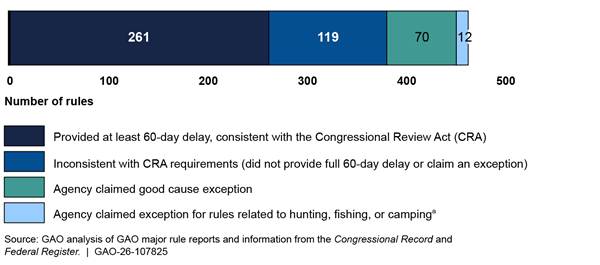
Notes: Two rules included a claim of good cause that covered part, but not all, of the rule, and the remaining parts had effective dates inconsistent with the CRA’s requirement. For purposes of this analysis, we counted these rules as inconsistent with CRA requirements and did not count them in the number of rules for which agencies claimed the good cause exception.
aThe CRA states that “any rule that establishes, modifies, opens, closes, or conducts a regulatory program for a commercial, recreational, or subsistence activity related to hunting, fishing, or camping . . . shall take effect at such time as the Federal agency promulgating the rule determines.” 5 U.S.C. § 808.
Most Rules with Inconsistent Effective Dates Provided Too Few Days Between Congressional Receipt and the Rule’s Effective Date
For most major rules that had stated effective dates inconsistent with the CRA, the date of receipt by Congress was less than 60 days before the published effective date of the rule.
Figure 5 shows the number of rules that had effective dates inconsistent with the CRA and whether this inconsistency was due to the date of receipt by Congress, the date of publication in the Federal Register, or both.[22]
|
Congressional Review Act 60-Day Delay Under the Congressional Review Act, unless an exception applies, major rules cannot take effect sooner than 60 days from the later of two dates: the date the Congress receives the rule and the date it is published in the Federal Register, if it is published. Source: 5 U.S.C. § 801(a)(3)(A). | GAO‑26‑107825 |
Figure 5: 119 Major Rules with Stated Effective Dates Inconsistent with the Congressional Review Act Because of Congressional Receipt Dates or Federal Register Publication Dates or Both, January 21, 2021, to January 20, 2025
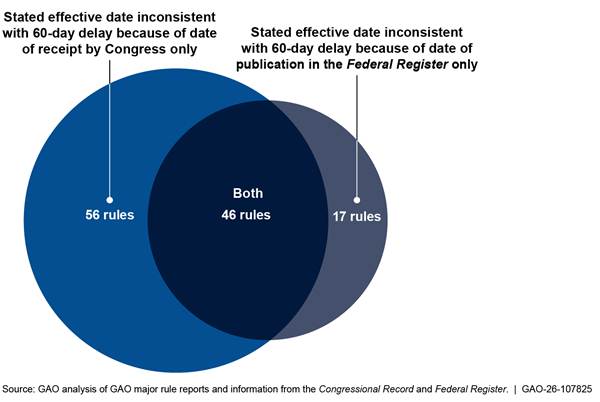
Later in this report, we analyze challenges agencies experienced in meeting CRA
requirements for the effective dates of major rules. For purposes of that
analysis, we review challenges with each of the CRA’s two effective date
requirements: (1) to allow at least 60 days after the date of publication in
the Federal Register and (2) to allow at least 60 days after the date
that Congress received the rule. For some rules, as shown in figure 5, agencies
may experience challenges with both requirements.
Agencies Provided Various Reasons when Claiming Good Cause for a Shorter Delay, Many Related to the COVID-19 Pandemic
Of the 462 major rules we reviewed, we identified 70 in which agencies claimed the good cause exception. As noted above, this exception allows agencies to set an effective date without a required delay.[23] As part of our analysis, we examined these rules and identified the following categories of explanations agencies provided (more than one category sometimes applies to a given rule):[24]
· the agency said it was responding to an emergency—31 rules, of which 28 were related to the COVID-19 pandemic;
· a law imposed a deadline either requiring the agency to issue a rule or requiring a program to be implemented by a date that agencies claimed would provide insufficient time to provide prior notice and comment—17 rules;
· a law prescribed the content of the rule issued—12 rules;
· a law required or authorized the agency to issue the rule as an interim final rule without providing notice or requesting public comment—12 rules;
· the statutory authority for issuing the rule was set to expire—4 rules; and
·
other explanations (for example, the rule made technical
corrections that an agency stated did not require public comment; or an agency
issued a rule in response to a court decision)—22 rules.
Inconsistency with the CRA’s 60-Day Delay Varied by Agency and by Year
Four Agencies Accounted for the Majority of Rules with Inconsistent Effective Dates
Our analysis found that 32 agencies issued at least one major rule from January 21, 2021, to January 20, 2025. Of these, the six that issued the greatest numbers of major rules accounted for about 55 percent of the 462 total major rules issued during this time frame. These agencies were HHS, the Environmental Protection Agency (EPA), the Securities and Exchange Commission (SEC), USDA, DOE, and Treasury. Four of these agencies collectively accounted for about 68 percent of the government-wide total of major rules with effective dates that were inconsistent with the CRA (81 of 119 major rules). The four agencies were: HHS (53 rules with dates inconsistent with the CRA), Treasury (13 rules), DOE (8 rules), and USDA (7 rules). The other two agencies—EPA and SEC—each issued one major rule with an effective date inconsistent with the CRA out of a total of 35 (EPA) and 29 (SEC) major rules issued during the time period.
HHS issued the greatest number of major rules (105), as well as the greatest number of major rules with effective dates inconsistent with the CRA (53). Furthermore, HHS issued a majority of the rules that were inconsistent with the CRA because of the Federal Register publication date. Of the total number of rules (63) that had Federal Register publication dates too close to the effective date, 44 were HHS rules, and most of those were issued by the Centers for Medicare & Medicaid Services. Figures 6 and 7 show the number of rules issued by HHS and other agencies during the Biden administration.
Figure 6: Major Rules Issued by the Department of Health and Human Services and Submitted to GAO, January 21, 2021, to January 20, 2025
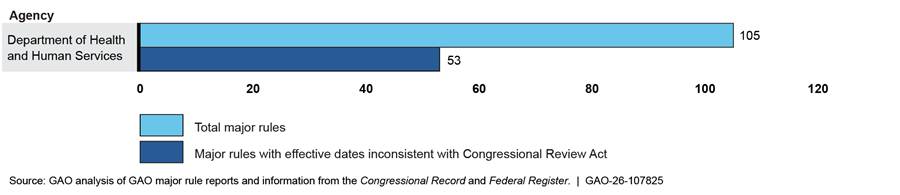
Note: Our scope included all major rules that were (1) published in the Federal Register from January 21, 2021, to January 20, 2025, and submitted to GAO for review under the Congressional Review Act (CRA); or (2) not published in the Federal Register but submitted to GAO for review under the CRA during the same period.
Figure 7: Major Rules Issued by Agencies Other Than the Department of Health and Human Services and Submitted to GAO, January 21, 2021, to January 20, 2025
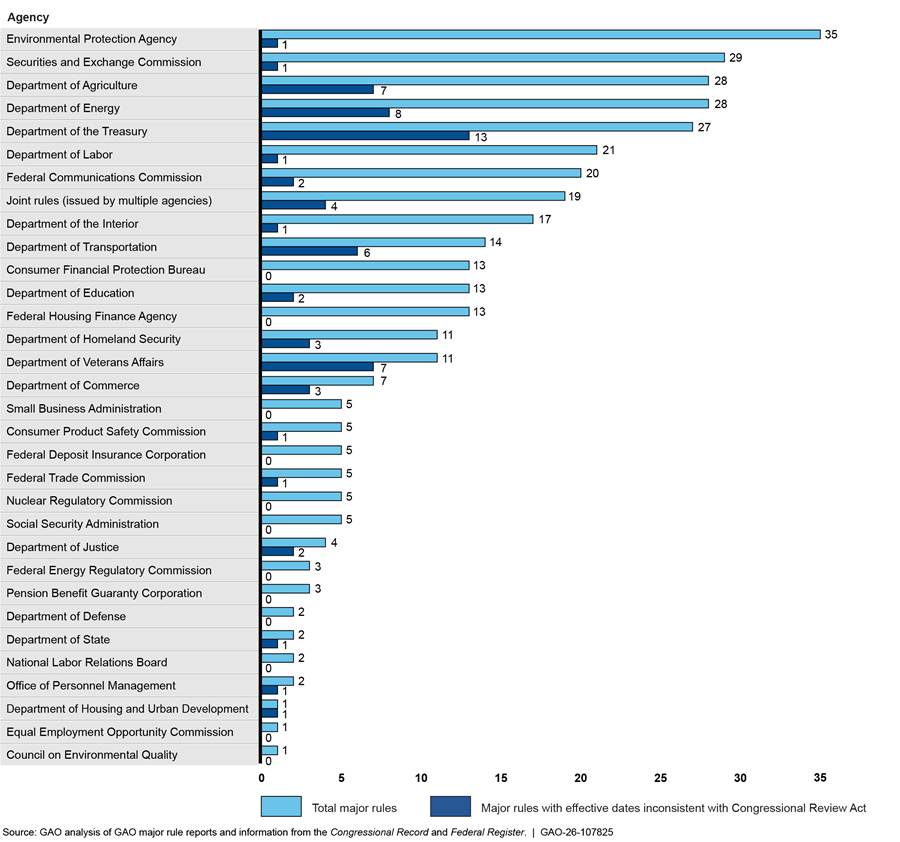
Note: Our scope included all major rules that were (1) published in the Federal Register from January 21, 2021, to January 20, 2025, and submitted to GAO for review under the Congressional Review Act (CRA), or (2) not published in the Federal Register but submitted to GAO for review under the CRA during the same period.
The Number of Rules with Inconsistent Effective Dates Varied by Year
The number of major rules with effective dates that were inconsistent with the CRA varied over the 4 years included in the scope of our review (see table 1). The percentage of rules issued that had effective dates inconsistent with the CRA was highest in the second year—January 21, 2022, through January 20, 2023. In that year, as table 1 shows, 27 of the 83 major rules issued (about 33 percent) had stated effective dates that did not provide the full 60-day delay and did not claim an exception to the delay requirement.
|
Time period |
Number of major rules issued |
Number of major rules with effective dates inconsistent with the Congressional Review Act (CRA) |
Percentage of major rules with effective dates inconsistent with the CRA |
|
Jan. 21, 2021–Jan. 20, 2022 |
93 |
19 |
20% |
|
Jan. 21, 2022–Jan. 20, 2023 |
83 |
27 |
33% |
|
Jan. 21, 2023–Jan. 20, 2024 |
101 |
31 |
31% |
|
Jan. 21, 2024–Jan. 20, 2025 |
185 |
42 |
23% |
|
Jan. 21, 2021–Jan. 20, 2025 (total) |
462 |
119 |
26% |
Source: GAO analysis of GAO major rule reports, Federal Rules System data, and information from the Congressional Record and Federal Register. I GAO‑26‑107825
Selected Agencies Reported Challenges Aligning Stated Effective Dates of Major Rules with CRA Requirements
While three of the four selected agencies—USDA, DOE, and Treasury—were generally able to ensure that major rules included an effective date that was at least 60 days after the date of publication in the Federal Register, HHS faced challenges doing so due to the types of rules HHS issues. In addition, all four selected agencies reported challenges related to delivering rules to Congress and understanding when their rules are considered received by Congress. These challenges contributed to these agencies’ difficulties in setting effective dates that were consistent with the 60-day delay.
While Most Selected Agencies Were Generally Able to Ensure a 60-Day Delay from the Federal Register Publication Date, HHS Faced Challenges
At three of the four selected agencies—USDA, DOE, and Treasury—most major rules included an effective date that was at least 60 days after the date of publication in the Federal Register. However, we found that the fourth agency, HHS, sometimes set effective dates for major rules in a way that necessarily provided less than the full 60-day delay from the date of publication in the Federal Register to the effective date, which is inconsistent with the CRA’s 60-day delay requirement. Among the four selected agencies, challenges with Federal Register publication were generally limited to HHS and were distinct from the challenges agencies experienced with ensuring timely receipt by Congress. Therefore, we discuss challenges related to receipt by Congress in a separate section below.
USDA, DOE, and Treasury issued a total of 67 major rules that were published in the Federal Register and did not claim an exception from the 60-day delay. Of those 67 rules, 61 delayed the stated effective date by at least 60 days from the Federal Register publication date. In contrast, HHS delayed the stated effective date by the required 60 days from the Federal Register publication date for about half of the rules that were published and did not claim an exception (43 of 87 rules).
|
Public Inspection Public inspection is a term the Office of the Federal Register uses to refer to displaying documents for the public to review before they are printed in the Federal Register. Rules posted for public inspection can be viewed on the Federal Register website. Source: GAO summary of Office of the Federal Register documents. | GAO‑26‑107825 |
OFR allows agencies to request a delayed effective date when submitting rules for publication. According to OFR officials, staff in that office calculate the rule’s effective date based on the submitting agency’s instructions once the publication date is known. For example, if an agency submitted a rule with the instructions, “This rule is effective [INSERT DATE 60 DAYS AFTER DATE OF PUBLICATION IN THE FEDERAL REGISTER],” and the rule was scheduled for publication on September 1, 2025, the final text of the rule would state, “This rule is effective on October 31, 2025.” If the calculated date falls on a weekend or holiday, the stated effective date would be the next business day. For example, a rule for which the agency specified a 60-day delay from the publication date would be effective 62 days from the publication date if the 60th day fell on a Saturday. Officials from USDA, DOE, and Treasury told us that one approach they use to ensure that they provide the 60-day delay is to instruct OFR to set the effective date of major rules 60 days from the date of publication in the Federal Register.
HHS officials told us that they, at times, also use this approach with the Federal Register publication date. However, they also stated that for some major rules, officials specify a different date calculation by instructing OFR to set the rule’s effective date 60 days from the date the rule is filed for public inspection, which occurs before the rule is officially published. This practice results in stated effective dates that are inconsistent with the CRA’s 60-day delay. Because a rule’s public inspection date precedes its Federal Register publication date, an effective date that is 60 days from the public inspection date will always fall short of providing the 60-day delay from publication specified in the CRA for major rules.
HHS officials told us that providing at least 60 days from the date of publication in the Federal Register is not always possible due to the types of rules HHS issues. Specifically, some lengthy and complex rules, such as those related to Medicare payments, must be made effective by certain dates because of statutory deadlines. However, according to HHS and OFR officials, processing and publishing these rules in the Federal Register has historically taken OFR weeks to complete due to their length and complexity. HHS officials told us that, for some of these rules, the agency bases the rule’s effective date on the public inspection date, which can be days or weeks before the rule’s publication date. That is, HHS sometimes delays effective dates by 60 days from the date a rule is first available for stakeholder review but not yet finalized and published in the Federal Register. However, this approach is not consistent with the CRA, which bases the 60-day delay on the Federal Register publication date and not the public inspection date.
OFR officials told us that, to reduce processing times for rules—including lengthier rules, such as those related to Medicare payments—OFR initiated a pilot program in June 2025.[25] As part of the pilot, OFR, among other things, eliminated some of its review steps and developed a government-wide template for agencies’ use in drafting the rule documents for OFR’s review. OFR officials told us that, according to the results of the pilot as of August 2025, the pilot reduced the average processing times for lengthier rules, such as HHS payment rules, which appeared as outliers in OFR’s initial review of rule processing times. HHS officials told us that, to the extent OFR’s pilot program maintains reduced processing times, HHS could consider setting effective dates based on their rules’ publication dates instead of their public inspection dates.
In the meantime, however, HHS has not leveraged other available options that could help ensure its rules are consistent with the CRA. For example, HHS may, in certain circumstances, be able to claim the CRA’s good cause exception. HHS officials told us that HHS’s Office of General Counsel assesses good cause on a case-by-case basis, based on facts and circumstances. They also stated that decisions about claiming good cause for rules within the scope of our review were made by senior officials in the prior presidential administration, and that they were not able to comment on the basis for those decisions. Further, HHS has the authority to make certain rules applicable retroactively, allowing the provisions of rules to apply before the effective date.[26] To the extent that HHS determines that these approaches are insufficient, HHS could seek legislative changes.
By using a rule’s public inspection date, rather than the Federal Register publication date, HHS routinely sets effective dates that fall short of providing the 60-day delay from the date of publication specified in the CRA. By taking steps to ensure its effective dates are consistent with the CRA, HHS would promote the CRA’s goal of providing a specific minimum delay in the effective dates of major rules to increase the chance that a rule’s disapproval, if any, would occur before the rule is implemented. Further, HHS would also reduce the risk of taking action on a rule before it goes into effect, such as by making larger Medicare payments.
Selected Agencies Faced Challenges Delivering Rules to Congress and Understanding When Congress Considers Them Received
All four selected agencies reported challenges related to delivering rules to Congress and understanding when their rules are considered received by Congress. These challenges contributed to the agencies’ difficulty in setting effective dates that were consistent with the 60-day delay. Our analysis of the four selected agencies’ major rules showed that these agencies did not consistently provide stated effective dates that were at least 60 days after the date Congress received their rules. For example, 65 of the 188 (35 percent) major rules we reviewed from the four agencies we selected did not provide the required 60-day delay after congressional receipt.
Challenges Ensuring Timely and Accurate Rule Delivery
Officials from all four selected agencies—USDA, DOE, HHS, and Treasury—told us that they experienced challenges with delivering rules to Congress. As described earlier, both the House and the Senate require agencies to submit paper copies of rules. Treasury officials told us they experienced delays when using the U.S. Postal Service and common carriers (e.g., FedEx or UPS) to mail rule reports to Congress. For example, Treasury officials told us that some providers attempted to deliver rules at times when Capitol buildings were closed, thereby delaying the delivery. USDA officials told us that routing the rules from the required drop-off location at the Capitol to the appropriate congressional office can take weeks in some cases, thereby contributing to delivery delays. Figure 8 shows how rules take different routes to the Capitol, depending on the delivery method agencies use.
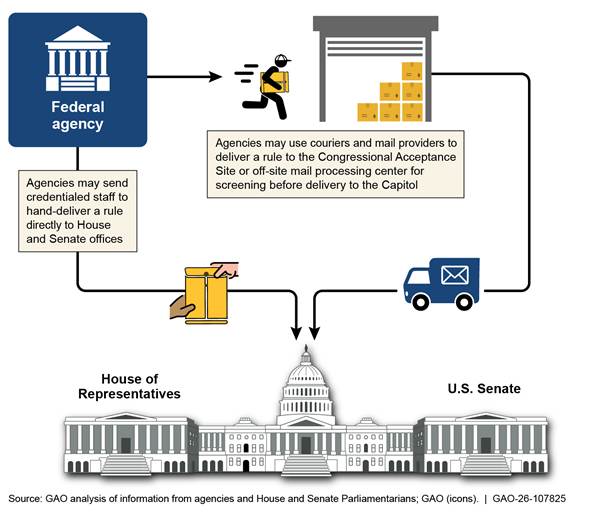
Note: This figure does not depict how rules are processed once delivered to the Capitol.
Officials from all four selected agencies told us that agency staff hand-deliver rules to the appropriate congressional offices. For example, a USDA official told us that from February 2024 to January 2025, USDA staff hand-delivered rules directly to House and Senate offices to help ensure timely processing and work around the delays of other delivery methods. However, some officials also noted that hand-delivering rules can be burdensome. For example, according to an HHS official, staff at HHS must hand-deliver every rule to Congress, following HHS’s standard operating procedures, and each trip to and from the Capitol typically takes an hour. Treasury officials also noted that frequent trips to the Capitol may not be an efficient use of their limited agency resources.
|
Parliamentarians’ Guidance for Agencies on Delivery of Rules According to the House Parliamentarian, for efficient processing in House offices, agencies’ reports on rules should be delivered directly to House Parliamentarian staff in the Speaker’s Rooms and must be provided in hard copy, with an ink signature, and addressed to the Speaker of the House. According to the Senate Parliamentarian, for efficient processing in Senate offices, reports on rules should be delivered to the office of the President of the Senate (Vice President of the United States). Source: Interviews with House and Senate Parliamentarians. | GAO‑26‑107825 |
While some selected agencies have specific instructions for where staff are to hand-deliver rules, Senate Parliamentarian staff told us that agency staff are, at times, unsure of the exact location for delivering rules, which contributed to delays. House Parliamentarian staff told us that rules are sometimes delivered to the main office of the Speaker of the House, rather than the Parliamentarian’s office within the Speaker’s floor office (see sidebar), delaying receipt because a rule is not considered received until it has been processed by the Parliamentarian staff on behalf of the Speaker. According to the Parliamentarian staff, people who work in the Speaker’s main office accept documents that are delivered there and determine which of them need to be brought to the Parliamentarian staff. By contrast, in the Senate, according to the Senate Parliamentarian, rules are not to be delivered directly to the Parliamentarian’s office, and doing so will cause delays, because the Parliamentarian staff will need to re-route the package to the Office of the President of the Senate.
Officials at all four selected agencies told us that electronically submitting rules to Congress would help resolve the challenges they experienced delivering rules by mail, courier, or by agency staff. In 2022, the Administrative Conference of the United States also recommended electronic submission of rules to Congress for CRA purposes.[27] However, the Senate Parliamentarian told us that implementing such a system would likely pose information security and policy challenges and incur significant maintenance costs. For purposes of this report, we did not assess the costs and benefits of changes to methods of delivery to Congress.
Uncertainty Concerning When Rules Are Considered Received by Congress
Once rules are delivered, agencies faced uncertainties about when rules are considered received by Congress. As shown in figure 9, rules are processed through offices in the House and the Senate before being referred to the committees of jurisdiction.
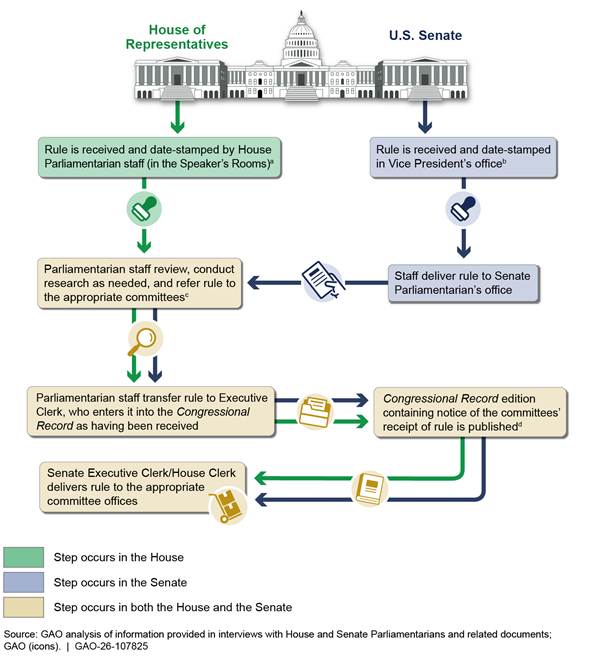
aAgencies may sometimes direct a rule to the Speaker of the House’s main office suite. The rule is then transferred to the House Parliamentarian for processing. According to the House Parliamentarian, a rule is considered received by the House on the date it is stamped by House Parliamentarian staff. This is the date shown in the Congressional Record.
bThe Congressional Record shows the date a rule was received in the Vice President’s Office.
cThe Parliamentarian staff refer rules to committees based on the subject matter of the rule and the jurisdiction of the committees.
dAccording to the Senate Parliamentarian, a rule is considered received by the Senate on the date notice of receipt is published in the Congressional Record. This is the date of publication, not the actual date of receipt stated in the notice.
The CRA requires rules to be received by Congress but does not further define what constitutes receipt by Congress or establish what must occur for the House and the Senate to receive a rule. Officials at selected agencies and at the House and Senate Parliamentarians’ offices described different ways they interpret the date that rules are to be considered received by Congress, and thus the CRA’s timing requirements. For example, USDA officials told us that they consider the date the rule is received by Congress as the date the rule is delivered to a mailroom in the Capitol. HHS officials told us that they consider the date an HHS employee hand-delivers the rule to congressional offices as the date the rule is received by Congress, but they are aware that Congress and GAO may interpret the date of receipt differently.
The Congressional Record typically indicates the date of receipt by the House of Representatives as the date the rule arrives in the House Parliamentarian’s office and the date of receipt by the Senate as the date it arrives in the Office of the President of the Senate. GAO’s major rule reports rely on these dates from the Congressional Record, when available, as the primary source for determining when Congress received a rule.[28] The Senate Parliamentarian told us that the Senate uses the publication date of the Congressional Record entry—not the actual date of receipt stated in the entry—as the date a rule is received for Senate procedural purposes.[29] House Parliamentarian staff stated that they consider a rule to be received by the House when it is stamped upon receipt in the Parliamentarian’s office.
Officials from DOE and Treasury said that increasing the amount of time they delay the stated effective date of a rule may help to address the uncertainty about Congress’s receipt of rules. For example, DOE officials told us they typically set the effective dates at 75 days from the date of publication in the Federal Register. However, DOE officials also told us that no department policy formalizes this practice.
Our analysis of rules issued during the Biden administration showed that DOE sometimes set effective dates for major rules 75 days from the date of publication in the Federal Register and that doing so appeared to facilitate meeting the requirements of the CRA in most but not all cases. Specifically, eight DOE rules had effective dates 75 (or 77 days including weekends) from the date of publication in the Federal Register, and all eight were consistent with the CRA’s 60-day delay requirement. By contrast, we found that for the nine DOE rules with stated effective dates 60 or 62 days after the date of publication, seven were inconsistent with the 60-day delay requirement because the stated effective date was less than 60 days after the rule was received by Congress. Of these seven rules, five would have been consistent with the CRA’s 60-day delay had the agency extended the delay between the Federal Register publication date and the rule’s effective date by an additional 15 days, to a total of 75 days.
A Treasury official told us that Treasury has considered adopting a 70-day delay for the effective dates of major rules to help ensure consistency with the CRA’s 60-day delay requirement, but it has not implemented this approach. According to Treasury and Internal Revenue Service (IRS) officials, taxpayers and tax practitioners rely on certain regulations issued by IRS and need them to take effect as soon as possible. We found that 15 Treasury rules had effective dates 60 to 62 days after the date of publication. Seven of these were consistent with the CRA’s 60-day delay requirement, and eight were inconsistent with the requirement (because of the date of receipt by Congress). All but one of the eight rules with inconsistent effective dates would have been consistent with the CRA’s delay requirement if the delay between the Federal Register publication date and effective date had been extended by 15 additional days.
Treasury staff also stated that during the COVID-19 pandemic emergency, IRS submitted rules to Congress using methods that did not provide delivery guarantees, such as the U.S. Postal Service and private mail services. Since the emergency ended, IRS has resumed delivering rules by hand. However, our analysis of major rules identified several rules with inconsistent effective dates that were issued after the COVID-19 emergency ended. For example, one IRS rule from 2024 related to partnership taxation provided 55 days between Congress’s receipt of the rule and its stated effective date.[30] When we asked about the potential benefits and challenges of adopting a standard delay longer than 60 days from Federal Register publication, Treasury and IRS officials said that adopting such a standard delay would interfere with taxpayers’ needs for certainty.
While asking OFR to calculate a later effective date to account for processing time in Congress may have addressed some challenges at DOE and Treasury, USDA and HHS faced a different set of challenges. At USDA, we identified seven rules with effective dates inconsistent with the 60-day delay. Three of these were initially not submitted to Congress because USDA did not consider them to be rules at the time they were issued.[31] The inconsistent effective dates for these rules were not caused by issues with delivery to Congress, but rather by differing legal interpretations about what is considered a rule. As discussed earlier, unlike the other three selected agencies, HHS faced challenges with using the Federal Register publication date. HHS officials told us that even if they were to use the Federal Register publication date to calculate the 60-day delay for major rules, this would not guarantee compliance with the CRA, because of uncertainty about what counts as the date Congress receives a rule. However, by increasing the delay after Federal Register publication to the 60 days specified in the CRA, HHS would also allow more time for processing in congressional offices than it currently does for some rules.
Under the CRA, major rules are generally required to take effect no sooner than 60 days after the rule is received by Congress or is published in the Federal Register, whichever date is later. As described earlier, agencies often attempt to comply with this delay requirement by delivering rules to Congress and instructing the Federal Register to delay the effective date by 60 days. However, agencies may in some cases fail to provide the full delay because of the uncertainties in predicting the date that Congress will report a rule as received. According to selected agency officials, there is often a difference of days or weeks between the date they deliver a rule to Congress and the date it is reported as received in the Congressional Record.
When agencies are uncertain about when a rule will be considered received by Congress, it limits their ability to determine a stated effective date that is consistent with the CRA. Officials from all four agencies we selected, which collectively accounted for about 68 percent of the total number of major rules with effective dates that were inconsistent with the CRA, told us that it is unclear to them how Congress determines the date of receipt that is published in the Congressional Record. According to some agency officials, this uncertainty makes it difficult to set effective dates when issuing rules. For example, HHS officials stated that without a clear definition of the date a rule is received by Congress that can be calculated at the time a rule is submitted for publication in the Federal Register, it is “virtually impossible” for HHS to comply with the CRA’s 60-day delay requirement. Congressional action is needed to reduce this uncertainty and promote the CRA’s goal of ensuring a specific minimum delay in the effective dates of major rules. The Senate Parliamentarian and the Deputy House Parliamentarian both expressed concerns about any change in the law that would affect dates used for procedural purposes in Congress. At the same time, they told us that the statutory provision that establishes the 60-day delay could be changed without changing the congressional procedure provisions of the CRA.
Conclusions
The 60-day waiting period for major rules is an important part of the CRA’s framework for congressional oversight of agency rulemaking. While Congress can use the CRA’s expedited procedures to disapprove a rule even after it has taken effect, the waiting period gives Congress a chance to do so before a rule’s effective date, potentially preventing it from being implemented. The CRA also provides exceptions that agencies can use in appropriate circumstances.
When agencies put rules into effect without waiting as long as the CRA requires and without claiming one of the exceptions Congress provided for in the act, two consequences may follow. First, Congress loses some or all of the time it would otherwise have to disapprove a rule before it takes effect. Second, agencies may take action on the basis of the rule earlier than allowed by law. Such actions could have material effects on the parties affected by the rule, such as changing the amount the government will pay providers for certain medical care.
While an agency controls when it sends a rule to Congress, it does not control the precise date Congress records the rule as “received.” As a result, agencies at times fail to allow the full waiting period because they must state an effective date when they issue a rule before knowing when the rule will count as having been received by Congress. Congress could address this challenge by giving agencies a clear, predictable standard regarding the receipt of rules. For example, the 60-day delay could start from a date that is within the agency’s control, such as the date the agency delivers copies of a rule to a specific congressional office or location.
At the same time, agencies can also improve their implementation of the CRA’s waiting period. When feasible, agencies can extend the effective date of a major rule beyond the minimum of 60 days, which can ensure that the rule is consistent with the CRA even when the precise timing of receipt by Congress is unknown. For example, DOE has been able to ensure consistency with the CRA for some rules by setting the effective date 75 days, instead of 60 days, from the date of publication in the Federal Register. Both DOE and Treasury could increase consistency with the CRA by adopting this approach for more rules. By contrast, HHS has often knowingly provided less than 60 days from the date of Federal Register publication by focusing on the date of public inspection. HHS would be better positioned to ensure consistency with the CRA by using other options to address challenges with statutory deadlines and the time it takes to publish lengthy, complex rules.
Matter for Congressional Consideration
Congress should consider amending the Congressional Review Act to specify when a rule is considered received by Congress for the purposes of section 801(a)(3)(A)(i), such as by stating that rules are considered received when they are delivered to a specific office or central delivery location for each House of Congress. (Matter for Consideration 1)
Recommendations for Executive Action
We are making a total of three recommendations, including one to HHS, one to DOE, and one to Treasury.
Specifically:
The Secretary of Health and Human Services should take steps to ensure the published effective date of rules is consistent with the CRA and other applicable statutory deadlines, in lieu of basing the effective date of the rule on the public inspection date. (Recommendation 1)
The Secretary of Energy should adopt a department-wide policy that, when feasible, major rules should be made effective at least 75 days after the date of publication in the Federal Register. (Recommendation 2)
The Secretary of the Treasury should adopt a department-wide policy that, when feasible, major rules should be made effective at least 75 days after the date of publication in the Federal Register. (Recommendation 3)
Agency Comments and Our Evaluation
We provided a draft of this report to USDA, DOE, HHS, Treasury, and the National Archives and Records Administration for their review and comment. USDA, DOE, and the National Archives and Records Administration stated they had no comments on the report.
Treasury’s Senior Legal Advisor for Regulatory Affairs provided comments via email, stating that Treasury agreed with our conclusions about amending the CRA to help allay the confusion agencies face with congressional processing of paper submissions and that Treasury intended to consider potential delays in congressional processing when determining the effective dates of major rules.
In its comments, reproduced in appendix II, HHS concurred with Recommendation 1, stating that establishing a general policy that supports compliance with the CRA is a good government practice. However, HHS also stated that establishing such a policy would not be sufficient to guarantee compliance with the CRA unless Congress establishes a common definition of “receipt” of a rule that is predictable at the time a rule is submitted for publication in the Federal Register. We agree that congressional action could help address uncertainty about when major rules are considered received by Congress for purposes of the CRA’s 60-day delay. At the same time, we believe that agencies, including HHS, can also improve their implementation of the 60-day delay through changes in their internal policies.
HHS also provided technical comments, which we incorporated as appropriate.
We are sending copies of this report to the appropriate congressional committees, the Secretary of Agriculture, the Secretary of Health and Human Services, the Secretary of Energy, the Secretary of the Treasury, and other interested parties. In addition, the report is available at no charge on the GAO website at http://www.gao.gov.
If you or your staff have any questions about this report, please contact me at JonesY@gao.gov. Contact points for our Offices of Congressional Relations and Media Relations may be found on the last page of this report. GAO staff who made key contributions to this report are listed in appendix III.

Yvonne D. Jones
Director, Strategic Issues
This report examines (1) the number of major rules issued from January 21, 2021, to January 20, 2025, that had effective dates inconsistent with the Congressional Review Act (CRA), (2) the distribution by agency and by year of rules with inconsistent effective dates, and (3) challenges selected agencies experienced in meeting CRA requirements for the effective dates of major rules.
To address our first objective, we reviewed the major rule report that GAO prepared for Congress for each of the 462 rules within the scope of our review. Our scope included all major rules that were (1) published in the Federal Register from January 21, 2021, to January 20, 2025, and submitted to GAO for review under the CRA, or (2) not published in the Federal Register but submitted to GAO for review under the CRA during the same period. This period represents the full term of the Biden administration.[32] We included rules regardless of whether they were later disapproved under the CRA or otherwise revoked, because our review focused on the effective dates that agencies set when issuing the rules. We did not evaluate the agencies’ decisions regarding procedural requirements or their determinations of the potential effects of their regulations—including determinations of which rules were major and therefore subject to the CRA’s 60-day delay.
We considered a rule to have a published effective date inconsistent with the CRA when any part of the rule met all three of the following conditions:
1. the agency did not claim the CRA’s exception for certain rules related to hunting, fishing, or camping;[33]
2. the agency did not claim the CRA’s good cause exception;[34] and
3. the stated effective date for any part of the rule was less than 60 days after either the date of receipt by the Senate, the date of receipt by the House of Representatives, or the date of publication in the Federal Register, if the rule was published in the Federal Register.[35]
Our analysis of the first and second conditions, regarding exceptions claimed by agencies, was based solely on the findings of GAO major rule reports. For two rules within our scope, the major rule report stated that the agency claimed good cause for part, but not all, of the rule. Because the remaining parts of these rules had effective dates inconsistent with the CRA, we counted these rules as having effective dates inconsistent with the CRA. For six rules within our scope, the agency attempted to claim the good cause exception, but we found in the major rule report that the good cause exception did not apply because the agency did not make a finding that notice and public procedure were impracticable, unnecessary, or contrary to the public interest. We did not count these as rules for which agencies claimed good cause, and we counted them as having effective dates inconsistent with the CRA when they also satisfied the other two conditions we identified.
For the third condition, we reviewed GAO major rule reports and, in certain circumstances, searched the Federal Register and the Congressional Record to identify changes that may have occurred after the major rule report was published.
To determine whether rules met the three conditions described above, a GAO analyst collected information about each rule based on the summary provided in the major rule report, including the date the rule was published in the Federal Register; the rule’s stated effective date; whether the stated effective date provided the 60-day delay; whether the agency claimed an exception from the delay requirement; and, for rules that did not provide the required delay or claim an exception, the date the rule was received by Congress. The major rule reports use the date recorded in the Congressional Record, when available, as the primary source for determining when Congress received a rule. When the Congressional Record has not published a notice of receipt within the 15-day time frame allotted for us to issue our major rule report, we accept evidence of delivery provided by the agency as establishing the date of receipt.
To validate the results of the analyst’s review, we completed an automated analysis of the major rule reports, using the same parameters. For the automated analysis, we programmed a script in R code to check for standard phrases that GAO includes in the text of major rule reports to describe exceptions claimed by agencies and identify inconsistencies between the stated effective date of the rule and the CRA’s 60-day delay. This automated analysis used two data sources: the full text of the major rule reports for the relevant period; and data from a separate database on rules that GAO maintains, the Federal Rules System.[36] Data we used from the Federal Rules System included key dates in the rules’ processing, including the effective date and Federal Register publication dates, as well as the name of the agency and subagency that submitted the rule.[37] For both sources, we used data as of July 18, 2025—the most recently available data at the time of our review. The latest-dated major rule report included in our analysis was issued on May 1, 2025, regarding a rule published in the Federal Register on January 17, 2025.[38] We compared the analyst-generated results with the automated results and resolved any discrepancies between the two.
GAO major rule reports are based on information available at the time they are issued, which the CRA specifies is to occur within 15 days after the date the rule is published in the Federal Register or received by Congress, whichever is later.[39] In some cases, relevant information about a rule may become available after the report is published, including information about receipt by Congress or agency-initiated changes to a rule’s effective date.
In two circumstances, we searched for such information and updated the results of our analysis based on relevant results, including the determinations of whether an agency had provided the 60-day delay. These circumstances included the following:
First, if the major rule report stated that the rule’s published effective date was inconsistent with the CRA, but the major rule report did not include the date the rule was received by Congress, we searched the Congressional Record for evidence of this date. Specifically, we used the date indicated in the Congressional Record as the date of receipt in the House or in the Senate, whichever is later. We used the later of the two dates because receipt by Congress requires receipt by both Houses. When we could not locate a relevant entry in the Congressional Record, we searched our CRA files for evidence provided by the agency. Such evidence included a delivery receipt showing the date the agency delivered the rule to the relevant House of Congress. We used the date documented in the receipt as evidence of the congressional receipt date. For six rules, we found evidence of receipt by one House of Congress, but not the other. In these instances, we recorded the date of receipt by the one House of Congress, which represents the earliest possible date of receipt by Congress.
Second, for the 132 rules that we identified as having stated effective dates inconsistent with the CRA, we searched the Federal Register to identify any changes agencies may have made to a rule’s effective date after it was published. Specifically, we searched for notices agencies published in the Federal Register that made the rule’s effective date consistent with the CRA’s 60-day delay. We found notices for 13 of the 132 rules. On the basis of the modified effective dates in these notices, we determined that the stated effective dates for these 13 rules were consistent with the CRA. We subtracted these 13 rules from the initial set of 132 rules we had initially identified as potentially inconsistent with the 60-day delay, which resulted in 119 rules that had stated effective dates that were inconsistent with the 60-day delay.
Based on this analysis, we assigned each rule to one of the following mutually exclusive categories: (1) rules that claimed the exception for certain rules related to hunting, fishing, or camping; (2) rules that claimed the good cause exception; (3) rules that provided at least a 60-day delay; and (4) rules that had effective dates inconsistent with the CRA. All rules where the agency claimed an exception were included in category (1) or (2), respectively, regardless of the number of days delay they provided. Category (2), for rules that claimed the good cause exception, did not include the two rules with good cause claims that only covered part of the rule, as discussed earlier, or the six rules for which agencies attempted to claim good cause, but GAO found and reported in the major rule report that the requirements of the statute were not satisfied.
For rules that we found were inconsistent with the CRA, we determined the number of days between the rule’s congressional receipt date and the rule’s stated effective date, and the number of days between the rule’s publication date in the Federal Register and the rule’s stated effective date.
To describe the reasons agencies provided when claiming the CRA’s good cause exception, we reviewed the text—as published in the Federal Register—of each of the 70 rules we identified as having claimed the good cause exception (not including the two rules with partial good cause claims or the six rules for which agencies attempted to claim good cause, but GAO found that the requirements of the statute were not satisfied). We developed the following categories of good cause:
· the agency said it was responding to an emergency;
· a law imposed a deadline either requiring the agency to issue a rule or requiring a program to be implemented by a date that agencies claimed would provide insufficient time to provide prior notice and comment;
· a law prescribed the content of the rule issued;
· a law required or authorized the agency to issue the rule as an interim final rule without providing notice or requesting public comment;
· the statutory authority for issuing the rule was set to expire; and
· other explanations (for example, the rule made technical corrections that an agency stated did not require public comment; or an agency issued a rule in response to a court decision).
To develop these categories, we initially used the list of categories from a previous report where we, among other things, analyzed the reasons agencies gave as the basis for findings of good cause for issuing final rules without a Notice of Proposed Rulemaking.[40] We added additional categories as appropriate based on the reasons agencies gave for good cause claims for the rules we reviewed in our scope. For example, we included a category for rules issued in response to the COVID-19 pandemic as a subcategory of the category on responding to emergencies. We assigned each rule to one or more categories according to the reasons the agency provided. This analysis was conducted by a GAO attorney and verified by a GAO analyst, resolving discrepancies as necessary. We did not separately evaluate whether each of these categories, taken as a whole, constituted a sufficient reason to claim good cause.
To assess the reliability of the data we analyzed for this work, which included the full text of GAO major rule reports and the Federal Rules System database, we reviewed prior data reliability assessments for these data sources, analyzed relevant documentation, tested for missing values, and compared the Federal Rules System data with the information in the text of the major rule reports. We determined that the data were sufficiently reliable for purposes of determining the numbers and key characteristics of rules under our review, such as dates and issuing agencies.
To address our second objective, we used the data produced through the analysis described above to identify the number of agencies that issued major rules during the January 21, 2021, to January 20, 2025, period, the number of rules each agency issued, and of those, the number that had stated effective dates inconsistent with the CRA, as well as the distribution of rules by year. For our analysis of rules by year, we used administration years, not calendar years. For example, we grouped together rules issued from January 21, 2021, to January 20, 2022.
We identified 462 major rules within our scope. For 443 of these rules, one of 32 agencies issued the rule. The remaining 19 rules were issued jointly by more than one agency. We treated these 19 joint rules as a separate category and did not attribute them to any one agency.
For the purposes of this report, we defined “agency” as a cabinet department or other agency with no higher-level parent agency, consistent with past GAO work. For example, we considered the Office of Personnel Management and the Department of the Treasury to be agencies. We categorized rules issued by components of an agency as issued by the agency. For example, we considered rules issued by the Financial Crimes Enforcement Network and the Internal Revenue Service as rules issued by Treasury.
To address our third objective, we selected the four agencies with the greatest number of rules that had stated effective dates inconsistent with the CRA’s 60-day delay requirement: the Departments of Agriculture (USDA), Energy (DOE), Health and Human Services (HHS), and the Treasury. For purposes of agency selection, we used data from the Federal Rules System and GAO major rule reports as of October 2024, the most recent data available at the time we initiated our review. We identified rules with stated effective dates inconsistent with the CRA using an automated analysis of those data.
We obtained and reviewed documentation of the four selected agencies’ policies and procedures for submitting rules to Congress, GAO, and the Federal Register, where available. USDA, HHS, and Treasury provided written documentation of procedures for submitting rules to Congress, and DOE did not. We interviewed officials responsible for carrying out these policies and procedures about any challenges their agencies may have experienced and potential solutions for addressing those challenges.
In addition, we interviewed the House and Senate Parliamentarians and their staff regarding procedures for receiving rules submitted by agencies. We selected these offices based on their prominent role in processing communications to Congress, including agency rules. We reviewed documents and interviewed officials from the Office of Management and Budget and National Archives and Records Administration regarding the requirements of the CRA and publication of rules in the Federal Register. Within the National Archives, the Office of the Federal Register is responsible for publishing the Federal Register, which includes proposed rules made available for public comment, final rules, and other documents from federal agencies, among other things. We spoke with officials in that office to obtain information about how agencies submit rules for publication and how rules are processed before publication.
We also interviewed knowledgeable individuals from three academic institutions, the Administrative Conference of the United States, and the Congressional Research Service to gain their insights about challenges related to meeting CRA requirements for the effective dates of rules.[41] We selected these individuals based on their publications and work experience related to the CRA.
We conducted this performance audit from September 2024 to April 2026 in accordance with generally accepted government auditing standards. Those standards require that we plan and perform the audit to obtain sufficient, appropriate evidence to provide a reasonable basis for our findings and conclusions based on our audit objectives. We believe that the evidence obtained provides a reasonable basis for our findings and conclusions based on our audit objectives.


Yvonne D. Jones, JonesY@gao.gov
In addition to the contact named above, Brenda Rabinowitz (Assistant Director), Theodore Alexander (Analyst-in-Charge), Michael Bechetti, John Bornmann, Leslie V. Gordon, Sheila R. McCoy, Jennifer Nguyen, Krista Loose, Charlotte McKiver, Danielle Novak, Douglas R. Sahmel, Andrew J. Stephens, Leanne V. Sullivan, and Emily Wilson Schwark made key contributions to this report.
The Government Accountability Office, the audit, evaluation, and investigative arm of Congress, exists to support Congress in meeting its constitutional responsibilities and to help improve the performance and accountability of the federal government for the American people. GAO examines the use of public funds; evaluates federal programs and policies; and provides analyses, recommendations, and other assistance to help Congress make informed oversight, policy, and funding decisions. GAO’s commitment to good government is reflected in its core values of accountability, integrity, and reliability.
Obtaining Copies of GAO Reports and Testimony
The fastest and easiest way to obtain copies of GAO documents at no cost is through our website. Each weekday afternoon, GAO posts on its website newly released reports, testimony, and correspondence. You can also subscribe to GAO’s email updates to receive notification of newly posted products.
Order by Phone
The price of each GAO publication reflects GAO’s actual cost of production and distribution and depends on the number of pages in the publication and whether the publication is printed in color or black and white. Pricing and ordering information is posted on GAO’s website, https://www.gao.gov/ordering.htm.
Place orders by calling (202) 512-6000, toll free (866) 801-7077,
or
TDD (202) 512-2537.
Orders may be paid for using American Express, Discover Card, MasterCard, Visa, check, or money order. Call for additional information.
Connect with GAO
Connect with GAO on X, LinkedIn, Instagram, and YouTube.
Subscribe to our Email Updates. Listen to our Podcasts.
Visit GAO on the web at https://www.gao.gov.
To Report Fraud, Waste, and Abuse in Federal Programs
Contact FraudNet:
Website: https://www.gao.gov/about/what-gao-does/fraudnet
Automated answering system: (800) 424-5454
Media Relations
Sarah Kaczmarek, Managing Director, Media@gao.gov
Congressional Relations
David A. Powner, Acting Managing Director, CongRel@gao.gov
General Inquiries
[1]Coast Guard, Department of Homeland Security, Safety Zone; New Year's Fireworks Show, Savannah River, Savannah, GA, 87 Fed. Reg. 80067 (Dec. 29, 2022).
[2]Federal Communications Commission, Facilitating Implementation of Next Generation 911 Services (NG911); Location-Based Routing for Wireless 911 Calls, 89 Fed. Reg. 78066 (Sep. 24, 2024); and Federal Communications Commission, Targeting and Eliminating Unlawful Text Messages, Implementation of the Telephone Consumer Protection Act of 1991, Advanced Methods to Target and Eliminate Unlawful Robocalls, 89 Fed. Reg. 5098 (Jan. 26, 2024).
[3]5 U.S.C. § 801(a)(3). The CRA defines major rules as follows: “[t]he term “major rule” means any rule that the Administrator of the Office of Information and Regulatory Affairs of the Office of Management and Budget finds has resulted in or is likely to result in—(A) an annual effect on the economy of $100,000,000 or more; (B) a major increase in costs or prices for consumers, individual industries, federal, state, or local government agencies, or geographic regions; or (C) significant adverse effects on competition, employment, investment, productivity, innovation, or on the ability of United States-based enterprises to compete with foreign-based enterprises in domestic and export markets. The term does not include any rule promulgated under the Telecommunications Act of 1996 and the amendments made by that Act.” 5 U.S.C. § 804(2).
[4]Statement for the Record by Senators Nickles, Reid, And Stevens, Subtitle E—Congressional Review Subtitle, 142 Cong. Rec. S3683, S3685 (April 18, 1996); Small Business Regulatory Enforcement Fairness Act, Views of the House Committees of Jurisdiction on the Congressional Intent Regarding the ‘‘Small Business Regulatory Enforcement Fairness Act of 1996,” 142 Cong. Rec. E571, E576 (April 19, 1996). The Small Business Regulatory Enforcement Fairness Act of 1996 is title II of the Contract with America Advancement Act of 1996; the CRA is subtitle E of that title. Pub. L. No. 104-121, § 251–253, 110 Stat. 847, 868–875 (1996).
[5]5 U.S.C. § 802.
[6]These numbers are as of April 6, 2026.
[7]5 U.S.C. § 808.
[8]GAO, Federal Rulemaking: Trends at the End of Presidents' Terms Remained Generally Consistent Across Administrations, GAO‑23‑105510 (Washington, D.C.: Jan. 31, 2023) and Federal Rulemaking: OMB Should Work with Agencies to Improve Congressional Review Act Compliance During and at the End of Presidents’ Terms, GAO‑18‑183 (Washington, D.C.: Mar. 13, 2018).
[9]Our scope included all major rules that were (1) published in the Federal Register from January 21, 2021, to January 20, 2025, and submitted to GAO for review under the CRA, or (2) not published in the Federal Register but submitted to GAO for review under the CRA during the same period.
[10]Our previous work reviewed agencies’ compliance with requirements of the federal rulemaking process, including the CRA, in selected transition and non-transition periods in the Clinton, Bush, Obama, and first Trump administrations. See GAO‑23‑105510; GAO‑18‑183.
[11]These reports can be found on our website: https://www.gao.gov/legal/congressional‑review‑act/reports‑on‑major‑rules. We prepare these reports to meet the CRA’s requirement that GAO report to Congress on whether an agency, in promulgating a major rule, has complied with certain procedural steps. 5 U.S.C. § 801(a)(2)(A). We do not analyze or comment on the substance or quality of rulemaking in these reports. These reports must be made to each House of Congress no later than 15 calendar days after a rule's submission to Congress or its publication in the Federal Register, whichever is later.
[12]The Administrative Conference of the United States is an independent federal agency composed of representatives from the public and private sectors to recommend improvements to administrative process and procedure to promote efficiency, participation, and fairness in the promulgation of federal regulations and in the administration of federal programs.
[13]5 U.S.C. § 802.
[14]5 U.S.C. § 801(a).
[15]The statutory delay generally supersedes any effective date the agency provides that is inconsistent with the CRA. 5 U.S.C. § 801(a)(3).
[16]5 U.S.C. § 801(a)(3). The CRA requires submission to both Houses of Congress. 5 U.S.C. § 801(a)(1)(A). Therefore, if the House and the Senate receive a rule on different dates, the date of receipt by Congress would be the later of the House receipt date and the Senate receipt date.
[17]The CRA states that “any rule that establishes, modifies, opens, closes, or conducts a regulatory program for a commercial, recreational, or subsistence activity related to hunting, fishing, or camping . . . shall take effect at such time as the Federal agency promulgating the rule determines.” 5 U.S.C. § 808. Other exceptions also exist; for instance, the CRA does not apply to Federal Reserve rules that concern monetary policy. 5 U.S.C. § 807.
[18]5 U.S.C. § 801(a)(2). See, for example, GAO, Federal Communications Commission: Facilitating Implementation of Next Generation 911 Services (NG911); Location-Based Routing for Wireless 911 Calls, B‑336722 (Washington, D.C.: Oct. 9, 2024).
[19]Proceedings of the House and the Senate are also recorded in the House Journal and Senate Journal, respectively. The Journals are the constitutionally mandated official records of the proceedings of the House and the Senate, and they record actions taken during each legislative day, including motions offered, votes taken, and amendments agreed to.
[20]In this report, we refer to rules that both failed to have the required 60-day delay and did not properly claim an exception as rules with effective dates inconsistent with the CRA, rules with effective dates inconsistent with the CRA’s 60-day delay, or rules with inconsistent effective dates. We use these terms interchangeably.
[21]This number does not include rules for which the agency claimed the good cause exception for part, but not all, of the rule (two rules). See, for example, GAO, Department of Health and Human Services, Centers for Medicare & Medicaid Services: Medicare Program; FY 2022 Hospice Wage Index and Payment Rate Update, Hospice Conditions of Participation Updates, Hospice and Home Health Quality Reporting Program Requirements, B‑333480 (Washington, D.C.: Aug. 18, 2021). It also does not include rules for which the agency attempted to claim the good cause exception, but we found in the major rule report that the agency had not satisfied the statutory requirement to include a finding that notice and public procedure on the rule were impracticable, unnecessary, or contrary to the public interest (six rules). See, for example, GAO, Department of Homeland Security, U.S. Citizenship and Immigration Services: Increase of the Automatic Extension Period of Employment Authorization and Documentation for Certain Employment Authorization Document Renewal Applicants, B‑336949 (Washington, D.C.: Jan. 3, 2025).
[22]To determine the date of receipt by Congress, we reviewed the GAO major rule report for each of these rules. When the major rule report did not contain complete information about the date of receipt by Congress because such information was not available at the time the major rule report was issued, we searched the Congressional Record, and, if needed, our internal CRA files, to fill in this missing information. See appendix I for more details on our methodology.
[23]The 70 rules do not include the two rules with claims of good cause that covered part, but not all, of the rule, and the six rules for which agencies attempted to claim the good cause exception, but GAO found in the major rule reports that the agency had not satisfied the statutory requirements for doing so.
[24]To develop these categories, we initially used the list of categories from a previous report: GAO, Federal Rulemaking: Agencies Could Take Additional Steps to Respond to Public Comments, GAO‑13‑21 (Washington, D.C.: Dec. 20, 2012). In that report, among other things, we analyzed the reasons agencies gave as the basis for findings of good cause for issuing final rules without a Notice of Proposed Rulemaking.
[25]The pilot program was created in response to Executive Order (EO) 14295, Increasing Efficiency at the Office of the Federal Register, 90 Fed. Reg. 20367 (May 9, 2025). EO 14295 requires the National Archives and Records Administration to reduce publication delays to the greatest extent feasible.
[26]The Medicare Prescription Drug, Improvement, and Modernization Act of 2003, Pub. L. No. 108-173, § 903, 117 Stat. 2066, 2376 (2003), codified at 42 U.S.C. § 1395hh(e)(1)(A), authorized the Secretary of Health and Human Services to make rules related to the administration of Medicare applicable retroactively when the Secretary determines that “(i) such retroactive application is necessary to comply with statutory requirements; or (ii) failure to apply the change retroactively would be contrary to the public interest.”
[27]Administrative Conference of the United States, Recommendation 2021-8, Technical Reform of the Congressional Review Act, 87 Fed. Reg. 1719 (Jan. 12, 2022).
[28]When the Congressional Record has not published a notice of receipt within the 15-day time frame we have to issue our major rule report, we accept evidence of delivery provided by the agency as establishing the date of receipt.
[29]The section of the CRA that establishes procedures and time frames related to resolutions of disapproval refers to the date rules are received by Congress. 5 U.S.C. § 802(b)(2)(A). The 60-day delay for major rules is established by a separate section of the statute, which also refers to the date rules are received by Congress. 5 U.S.C. § 801(a)(3)(A)(i).
[30]89 Fed. Reg. 91552 (Nov. 20, 2024). See GAO, Department of the Treasury, Internal Revenue Service: Election to Exclude Certain Unincorporated Organizations Owned by Applicable Entities from Application of the Rules on Partners and Partnerships, B‑336883 (Washington, D.C.: Dec. 10, 2024).
[31]These rules were grant-related announcements that were not published in the Federal Register, and USDA officials told us that agency leadership did not consider such announcements to be rules for purposes of the CRA. In June 2023, in response to a request from a Member of Congress, we issued a legal opinion that concluded that certain grant-related documents were rules. GAO, U.S. Department of Agriculture—Applicability of the Congressional Review Act to Documents Implementing Four Financial Assistance Programs, B‑334146 (Washington, D.C.: June 5, 2023). According to USDA officials, USDA submitted previously issued funding announcements to GAO and Congress in response to the legal opinion.
[32]Our previous work reviewed agencies’ compliance with requirements of the federal rulemaking process, including the CRA, in selected transition and nontransition periods in the Clinton, Bush, Obama, and first Trump administrations. See GAO‑23‑105510 and GAO‑18‑183.
[33]5 U.S.C. § 808(1).
[34]5 U.S.C. § 808(2).
[35]5 U.S.C. § 801(a)(3). A rule may have multiple effective dates, for example when amendments to different sections of the Code of Federal Regulations are to take effect at different times. In such cases, we considered the rule to have a published effective date inconsistent with the CRA if any one or more of its stated effective dates were inconsistent with the CRA.
[36]Our website includes a public database of rules submitted to us, both major and non-major. GAO, “Search Database of Rules,” accessed December 31, 2025, https://www.gao.gov/legal/congressional‑review‑act/search‑database‑of‑rules.
[37]We refer to a component of a larger department-level agency as a subagency.
[38]GAO, Department of Homeland Security, United States Coast Guard: Cybersecurity in the Marine Transportation System, B-337400 (Washington, D.C.: May 1, 2025).
[39]5 U.S.C. § 801(a)(2)(A).
[40]GAO, Federal Rulemaking: Agencies Could Take Additional Steps to Respond to Public Comments, GAO‑13‑21 (Washington, D.C.: Dec. 20, 2012).
[41]The Administrative Conference of the United States is an independent federal agency composed of representatives from the public and private sectors to recommend improvements to administrative process and procedure to promote efficiency, participation, and fairness in the promulgation of federal regulations and in the administration of federal programs.
