FDA ADVISORY COMMITTEES
More Transparency Needed on Policies for Making Conflict of Interest Determinations
Report to Congressional Committees
United States Government Accountability Office
A report to congressional committees.
For more information, contact: Mary Denigan-Macauley at deniganmacauleym@gao.gov
What GAO Found
The Food and Drug Administration (FDA) relies on more than 30 advisory committees to provide expert advice on numerous scientific topics, including opioid drugs for pain. Government-wide requirements prohibit committee members from taking part in meetings that could affect their financial interest. FDA checks for conflicts of interest by reviewing a form that committee members fill out to disclose financial interests, such as investments in drug companies. Regulations also require FDA to check for issues that could give the appearance that a committee member lacks impartiality. In some situations, FDA may grant a waiver or authorize a member to take part in a meeting, such as when the need for the member’s expertise outweighs the potential conflict. FDA also has policies to similarly review financial interests and appearance issues for guest speakers, who may be invited to meetings to present scientific information.

Between June 2018 and May 2025, FDA invited an average of about 29 committee members to each of the 17 most recent meetings for committees that discuss opioids. Across these meetings, FDA’s conflict of interest review process resulted in members being recused from taking part in the meetings 15 times. FDA also granted a financial conflict of interest waiver to allow a member to participate because the agency determined their expertise was needed and that their financial interest was not substantial. In addition, FDA authorized members with appearance issues to take part in meetings seven times. These results are comparable to those of other FDA committees that GAO reviewed.
FDA uses a combination of government-wide requirements and internal policies to guide its conflict of interest review process. However, FDA does not publicly share information on how it determines whether members have financial conflicts of interest and whether they should participate in committee meetings. This is because FDA has not yet finalized required guidance on the matter more than 13 years after a law required it. In addition, FDA has not posted to its website how it makes these decisions in the interim. Further, the agency does not publicly share how it determines whether guest speakers have financial conflicts or appearance issues and whether they should participate in meetings. Publicly sharing information on how FDA makes these determinations would be consistent with the law and FDA’s own best practices. Making this information public would increase transparency and provide the public with greater assurance that FDA has steps in place to manage conflicts of interest for advisory committees, and therefore help to ensure accountability and consistency in decision making.
Why GAO Did This Study
Conflict of interest rules and guidelines are essential to ensure FDA receives independent, unbiased professional expertise from advisory committees to help its efforts to assure the safety and effectiveness of drugs, medical devices, and other products. Federal requirements for advisory committees emphasize the importance of public access, input, and accountability, including addressing conflicts of interest.
A Senate Appropriations Committee report includes a provision for GAO to review how FDA addresses conflicts of interest for advisory committee meetings, particularly for opioids.
This report (1) describes FDA’s advisory committee conflict of interest policies and review process, (2) describes the results of FDA’s review process for advisory committees related to opioids, and (3) examines the extent to which FDA publicly shares its conflict of interest policies for advisory committees.
GAO reviewed relevant federal laws and regulations, as well as FDA documentation on advisory committees. GAO also reviewed published research about the results of FDA’s conflict of interest review process and interviewed FDA officials and stakeholders familiar with advisory committee meetings.
What GAO Recommends
GAO is recommending that FDA (1) establish a time frame
for issuing and publicly sharing required financial
conflict of interest guidance, (2) make public how it determines these conflicts
for committee members in the interim, and (3) make public how it
determines conflicts and appearance issues for guest speakers.
HHS concurred with the recommendations.
Abbreviations
AADPAC Anesthetic and
Analgesic Drug Products Advisory
Committee
CDER Center for Drug Evaluation and Research
DSaRM Drug Safety and Risk Management Advisory Committee
FDA Food and Drug Administration
HHS Department of Health and Human Services
This is a work of the U.S. government and is not subject to copyright protection in the United States. The published product may be reproduced and distributed in its entirety without further permission from GAO. However, because this work may contain copyrighted images or other material, permission from the copyright holder may be necessary if you wish to reproduce this material separately.
March 24, 2026
The Honorable John Hoeven
Chair
The Honorable Jeanne Shaheen
Ranking Member
Subcommittee on Agriculture, Rural Development, Food and Drug Administration,
and Related Agencies
Committee on Appropriations
United States Senate
The Honorable Andy Harris
Chairman
The Honorable Sanford Bishop, Jr.
Ranking Member
Subcommittee on Agriculture, Rural Development, Food and Drug Administration,
and Related Agencies
Committee on Appropriations
House of Representatives
Advisory committees are a useful tool for furnishing expert advice, ideas, and diverse opinions to the federal government, as characterized by the Federal Advisory Committee Act, and they operate under transparency laws that aim to promote public access, input, and accountability.[1] The Food and Drug Administration (FDA) uses advisory committees to provide independent expertise and technical assistance to help the agency make decisions about the development and evaluation of drugs and other products.
FDA convenes advisory committee meetings publicly to discuss and evaluate information on a specific topic, such as a pharmaceutical company’s application to market a new cancer drug. These meetings involve committee members appointed by FDA, and may involve individuals who speak on behalf of companies whose products are being discussed, or guest speakers, such as researchers who are invited to present scientific papers. Members of the public with relevant information or views are also allowed to speak during certain portions of the meetings.
Committee members and other meeting participants may have potential conflicts of interest, such as financial investments in drug manufacturers, or relationships with companies that could give the appearance of a conflict.[2] Federal law requires federal agencies, including FDA, to address potential conflicts of interest that may affect advisory committees.[3] In 2008, we described FDA’s process for evaluating potential conflicts of interest, including situations when committee members are prohibited from participating in meetings, and circumstances when members with conflicts may be allowed to participate.[4] Additionally, in 2020, we reviewed 11 advisory committees across multiple federal agencies, including FDA, and found that they met many, but not all selected transparency requirements established by federal law and regulations.[5] We made nine recommendations, all of which federal agencies implemented as of January 2025.[6]
In an April 2025 press release, the FDA Commissioner announced that FDA would, where statutorily permitted, remove advisory committee members who represent industry to mitigate perceived industry influence and to be consistent with a Department of Health and Human Services (HHS)-wide goal of increasing transparency.[7] In the announcement, the Commissioner stated that there were low levels of public trust, and emphasized the need to avoid potential conflicts of interest.
Additionally, recent Senate Appropriations Committee reports raised questions about the possibility of inappropriate industry influence on FDA advisory committees, particularly for opioid painkillers. A report accompanying the Consolidated Appropriations Act, 2024, includes a provision for us to review how FDA addresses conflicts of interest for advisory committees.[8]
In this report, we:
1. Describe FDA’s conflict of interest policies and related review process for advisory committees.
2. Describe the results of FDA’s conflict of interest review process for advisory committees that review opioids.
3. Examine the extent to which FDA publicly shares its conflict of interest policies for advisory committees.
To describe FDA’s policies and process for addressing conflicts of interest for advisory committees, we reviewed federal statutes and regulations and agency documentation. We also interviewed FDA officials about how they review advisory committee participants’ disclosed interests for conflicts.[9] We focused on conflicts of interest as they relate to participation in advisory committee meetings, and did not review other policies and processes, such as appointing or dismissing advisory committee members, or creating or dissolving advisory committees. In addition, we did not review FDA expert panels, which according to FDA officials are different from advisory committees and not subject to the Federal Advisory Committee Act.[10]
To describe the results of FDA’s conflict of interest processes for advisory committees that review opioids, we reviewed publicly available meeting documentation. Documentation included meeting agendas and transcripts for the 17 committee meetings that took place between June 2018 and May 2025. We focused on the committees that review opioids due to the ongoing opioid crisis. A public health emergency declaration for the opioid crisis has been in effect since October 2017, due to high rates of opioid use disorder and related deaths.[11] Additionally, questions have been raised about the possibility of inappropriate industry influence on FDA advisory committees, particularly for opioid painkillers.[12] We selected the time frame to include a variety of recent advisory committee meetings across three administrations. To provide context, we compared the results of FDA’s conflict of interest review process for committees that review opioids with available information on other FDA committees.
We reviewed FDA’s annual reports for fiscal years 2018 through 2024, which contain information on certain results of FDA’s conflict of interest processes for each advisory committee meeting. According to FDA’s annual report from fiscal year 2024, the most recent report available at the time of our review, FDA determined that some reports included a limited amount of inaccurate information. To assess the reliability of data we used from FDA annual reports, we cross-checked data with documentation available on FDA’s advisory committee website, when possible. We also interviewed agency officials with knowledge about these data to confirm that aggregate numbers presented in annual reports are accurate and complete, to confirm that our information sources and methodology were appropriate, and to identify any limitations. As a result of these steps, we determined that the data were sufficiently reliable for the purposes of our reporting objectives.
To examine the extent to which FDA publicly shares its conflicts of interest policies for advisory committees, we reviewed FDA’s advisory committee website, which included information on relevant laws, regulations, and guidance.[13] We also examined the extent to which FDA shared information with the public about its policies for addressing advisory committee conflicts of interest compared to (1) federal regulations that call for agencies to create publicly facing advisory committee websites, (2) requirements under the Federal Food, Drug, and Cosmetic Act for FDA to issue guidance on how it reviews certain financial conflicts of interest for committee members, (3) FDA’s regulations on good guidance practices, particularly the practice of publicly sharing guidance documents, and (4) FDA’s 2024 Report and Plan on Best Practices for Guidance.
To inform the second and third objectives, we conducted a literature review of relevant peer-reviewed research published in the last 20 years. Specifically, we identified and reviewed 16 articles that studied conflicts of interest among various FDA advisory committee participants. We also interviewed representatives from a nongeneralizable selection of 10 research and public interest groups (stakeholders) about their knowledge of, and experience with FDA’s advisory committees. These stakeholders spoke about FDA advisory committees in general, and their comments were not specific to the committees that review opioids. We identified potential stakeholders to interview from among individuals and organizations with relevant knowledge or perspectives who (1) submitted comments to FDA on advisory committees, (2) published articles on FDA advisory committees, or (3) were referred to us by other stakeholders. (See appendix I for more information on stakeholder selection.)
We conducted this performance audit from October 2024 to March 2026 in accordance with generally accepted government auditing standards. Those standards require that we plan and perform the audit to obtain sufficient, appropriate evidence to provide a reasonable basis for our findings and conclusions based on our audit objectives. We believe that the evidence obtained provides a reasonable basis for our findings and conclusions based on our objectives.
Background
|
Food and Drug Administration (FDA) Centers with Advisory Committees Four of FDA’s centers convene advisory committees, which provide expertise and technical assistance to help the agency make decisions about the development and evaluation of products regulated by FDA: · Center for Biologics Evaluation and Research regulates biological products for human use such as blood, tissue, and vaccines. · Center for Devices and Radiological Health regulates medical devices such as pacemakers and blood glucose monitors. · Center for Drug Evaluation and Research regulates prescription and over-the-counter drugs. · Center for Tobacco Products regulates the manufacturing, marketing, and distribution of tobacco products. Source: GAO analysis of FDA documentation. | GAO‑26‑107877 |
FDA has approximately 30 advisory committees across its centers, as well as its Office of the Commissioner, covering topics such as drug evaluation and medical devices.[14]
FDA has two committees that review opioids—the Anesthetic and Analgesic Drug Products Advisory Committee (AADPAC) and the Drug Safety and Risk Management Advisory Committee (DSaRM), both of which are within the Center for Drug Evaluation and Research (CDER). The charters for these committees specify that each is to have 11 core voting members, including one member representing consumer interests. Advisory committees often vote on questions, such as whether FDA should approve a new drug application. FDA takes into consideration recommendations made by advisory committees, including deliberations and voting, but the agency makes final decisions. In addition to voting members, these committees may also include one non-voting member who represents industry interests and is not subject to conflict of interest requirements. According to the charters, AADPAC members may be experts in anesthesiology, analgesics (such as issues related to opioid abuse), epidemiology, or statistics. DSaRM members are to be knowledgeable in the fields of risk communication, risk management, drug safety, and issues related to drug abuse.
Most members of FDA advisory committees are standing members, who serve terms of up to 4 years, unless they resign or are removed from the membership by the Secretary of Health and Human Services or FDA Commissioner.[15] Standing committee members generally are appointed as special government employees.[16] FDA may also appoint temporary members to serve for a specific advisory committee meeting, such as experts in the specific topic being discussed at the meeting. FDA generally follows a nomination process to appoint members, which includes publishing a request for nominations in the Federal Register.[17] However, the Secretary of Health and Human Services, the Assistant Secretary for Health, or the FDA Commissioner may also appoint members, according to FDA regulations.[18]
FDA Uses a Combination of Government-Wide Requirements and Internal Policies to Guide Its Conflict of Interest Review Process
Conflict of Interest Requirements and Review Process for FDA Advisory Committee Members
FDA uses federal statutes and Office of Government Ethics regulations to guide its review of financial interests for advisory committee members, and recuse—that is, exclude—members who FDA has determined have a conflict of interest from participating in committee meetings.[19] Federal law generally prohibits federal employees, including special government employees, from participating in advisory committee meetings that could affect the employee’s financial interests or the interests of others, such as spouses and children.[20]
In addition to financial conflicts of interest, FDA’s review also considers whether a committee member’s interests or relationships create the appearance that the member lacks impartiality on the issue being considered.[21] For example, the appearance of a conflict of interest may occur when an advisory committee member has a close relationship with one of the parties involved in the meeting, or with someone who has a financial interest that could be affected by the committee’s actions. FDA refers to the appearance of a conflict as an “appearance issue.”
FDA requires committee members to complete FDA’s Confidential Financial Disclosure Report for Special Government Employees in advance of committee meetings. The disclosure report requests information such as
· The member’s current financial interests, as well as those of their spouse, dependent child, or general partner in a business organization (e.g., investments, bonds, retirement plans, contracts, and grants) that have a link to the meeting task or topic, and
· Other relationships that have a link to the meeting task or topic that may create an appearance of a conflict.[22] (See Fig. 1.)
Figure 1: Examples of Financial Interests the Food and Drug Administration (FDA) Reviews for Advisory Committee Meetings

According to FDA officials, FDA staff review the disclosures to determine whether members have a financial conflict of interest or the appearance that they lack impartiality.[23] If FDA staff determine that a committee member has a financial conflict or appearance issue, they choose between two options
1. Recuse the member from participating, or accept the member’s own decision to recuse themselves (with or without FDA seeking a replacement), or
2. Decide the member’s expertise is needed and that the member meets the criteria for a financial conflict of interest waiver or appearance authorization.[24]
Federal agencies, including FDA, have authority to allow committee members with financial conflicts to participate in committee meetings under certain circumstances. Specifically, a federal agency may grant a waiver for a financial conflict of interest if it determines the interest is not so substantial that it would likely affect the integrity of the services that may be expected from the committee member.[25] Federal agencies may also grant a financial conflict of interest waiver if the agency determines that the need for the committee member’s services outweighs the potential for a conflict of interest.[26] Federal agencies are generally required to make conflict of interest waivers available to the public.[27]
Similarly, when a member has an appearance issue, a federal agency may determine that the government’s interest in the member’s participation outweighs the concern that a reasonable person may question the member’s impartiality. In these cases, the federal agency may grant an appearance authorization to allow a committee member to participate in the meeting.[28] An appearance authorization is similar to a financial conflict of interest waiver but is allowed by different legal authorities. (See table 1.)
|
Authority |
Criteria |
|
Statutory waivers |
|
|
18 U.S.C. § 208(b)(3) waiver of the criminal financial conflict of interest specific to special government employees serving on an advisory committee subject to the Federal Advisory Committee Act |
The need for the committee member’s service outweighs the potential for a conflict of interest created by the financial interest involved. |
|
18 U.S.C. § 208(b)(1) waiver of the criminal financial conflict of interest |
The financial interest involved is not so substantial as to be deemed likely to affect the integrity of the services which the government may expect from the committee member. |
|
Regulatory authorization |
|
|
5 C.F.R. § 2635.502 appearance authorization to allow a committee member to participate when their impartiality could be questioned |
The government’s interests in the member’s participation outweigh the concern that a reasonable person may question the integrity of the agency’s programs and operations. |
Source: GAO analysis of applicable conflict of interest statute and regulation. | GAO‑26‑107877
In addition to government-wide conflict of interest requirements, FDA also has agency-specific requirements regarding the public disclosure of advisory committee members’ financial waivers, as specified in the Federal Food, Drug, and Cosmetic Act. If FDA grants a waiver to allow a committee member with a conflict to participate in a meeting, the act requires the agency to publicly disclose the type, nature, and magnitude of the member’s financial interest no later than 15 days prior to the meeting.[29] The act also requires FDA to disclose the reasons for the waiver, including, as appropriate, the public health interest in having the member’s expertise on the particular topic before the committee.[30]
As part of its disclosure process, FDA posts financial conflict of interest waivers on its website. Additionally, according to FDA guidance the agency makes a disclosure statement on the details of any financial waivers during advisory committee meetings. FDA documents these disclosure statements in meeting transcripts, which become publicly available within 90 days after the meeting, according to FDA’s website. The agency does not disclose similar information about appearance authorizations, as doing so is generally prohibited under regulations issued by the Office of Government Ethics, according to FDA officials.[31]
|
Example of Food and Drug Administration (FDA) Advisory Committee Conflict of Interest Review Process For advisory committees within FDA’s Center for Drug Evaluation and Research (CDER), FDA staff from within the center determine the topic and expertise needed for an advisory committee meeting. As of March 2026, staff from the FDA Office of Ethics and Integrity review committee members’ financial disclosure forms to determine if a member has a financial conflict of interest or appearance issue. If they determine a member has a financial conflict of interest or appearance issue, then they work with CDER staff to determine whether to recuse the member, or whether to seek a waiver or authorization that would allow the member to participate. To pursue a waiver or authorization—when they determine that the member’s expertise needed for the meeting outweighs the conflict, or the conflict is not substantial—they work with FDA Advisory Committee Oversight and Management staff to obtain the necessary reviews and approval. Advisory Committee Oversight and Management staff work with all FDA centers to help ensure consistent operations within FDA’s advisory committee program, according to an internal staff manual guide. Additionally, Advisory Committee Oversight and Management staff serve to help ensure FDA advisory committee activities are consistent with the Federal Advisory Committee Act and other federal laws and agency policies. |
Source: GAO analysis of FDA internal documentation and interviews with FDA officials. | GAO‑26‑107877
|
Food and Drug Administration (FDA) Advisory Committee Conflict of Interest Guidance FDA has some final and draft guidance documents for staff, the public, and advisory committee members related to different aspects of conflict of interest: · Procedures for Evaluating Appearance Issues and Granting Authorizations for Participation in FDA Advisory Committees (draft June 2016), which describes FDA’s process for: · Evaluating a committee member’s financial interests and relationships for potential appearance issues, and · Granting an appearance authorization. · Public Availability of Advisory Committee Members’ Financial Interest Information and Waivers (final March 2014), which describes FDA’s process for: · Publicly disclosing financial interests and waivers for committee members. · The Open Public Hearing at FDA Advisory Committee Meetings (final May 2013), which describes FDA’s process for: · Public participation in advisory committee meetings, and · Encouraging public speakers to disclose relevant financial relationships. Source: GAO analysis of FDA documentation. | GAO‑26‑107877 |
FDA also has some guidance documents that detail how it addresses types of conflict of interest and appearance issues for advisory committees. These guidance documents describe FDA policies on evaluating appearance issues for advisory committee members and the public availability of committee members’ financial interest information. (See sidebar for more information.) FDA’s guidance documents describe the agency’s current thinking on a topic, but do not establish legally enforceable responsibilities, according to the agency.
FDA Conflict of Interest Policies and Review Process for Advisory Committee Guest Speakers
FDA has agency-specific policies for guest speakers at advisory committee meetings. Specifically, FDA developed an internal staff manual on how FDA staff should determine whether these speakers have conflicts of interest. According to FDA’s staff manual, FDA may choose to invite guest speakers, and they are expected to have special professional, technical, or scientific information relative to the matters under consideration by the advisory committee. Similar to committee members, FDA requires guest speakers to disclose any financial interests or professional relationships relevant to the committee meeting, as a condition of invitation.[32] FDA requires guest speakers to complete a Financial Interest and Professional Relationship Certification for Guests and Guest Speakers form, which asks for the same type of information that the advisory committee member disclosure requests.
If FDA determines that a speaker has a financial conflict or appearance issue, the agency may rescind the invitation to speak, according to the FDA staff manual guide. While this is similar to FDA’s conflict of interest review process for committee members, FDA officials may choose to allow a guest speaker to participate in a committee meeting without the need for a formal waiver or authorization, according to FDA officials. In that case, the manual states that a guest speaker must acknowledge their interests either at the beginning of the meeting or at the beginning of their presentation, and this acknowledgement will be recorded into the meeting transcript.
FDA’s Review Process for Opioid Committees Resulted in Recusals, a Waiver, and Public Disclosures
FDA held 17 meetings for AADPAC and DSaRM, the two committees that review opioids, between June 2018 and May 2025. Among other things, the meetings considered public health risks related to the misuse or accidental exposure to opioids, as well as data on the safety and effectiveness of these drugs.[33] FDA’s conflict of interest review process for these meetings resulted in recusals, a financial waiver, appearance authorizations, and public disclosures of financial interests or conflicts. These results applied to FDA’s review process for advisory committee members and guest speakers who participated in these committee meetings. We also reviewed disclosures made by open public hearing speakers present at these meetings.[34]
Advisory Committee Members
According to FDA’s annual reports on advisory committees, the agency invited an average of about 29 advisory committee members across the 17 meetings held by AADPAC and DSaRM between June 2018 and May 2025, for a total of 496 invitations. In total, members were recused from participation 15 times. (See table 2.) In addition, FDA’s annual reports indicate that these results are comparable to meetings for the other 17 CDER committees (including any joint meetings held with DSaRM) that took place within this time frame. Between June 2018 and the end of fiscal year 2024, FDA’s annual reports indicate two other CDER advisory committees also made at least 15 recusals.[35] The remaining 15 CDER committees recused members between zero and 11 times. These recusals were based on either the member’s choice (a self-recusal) or the agency’s determination that a member had a conflict of interest related to a given meeting’s topic.[36]
Table 2: Results of the Food and Drug Administration’s (FDA) Conflict of Interest Review Process for Opioid-Related Advisory Committees from June 2018 to May 2025
|
Number of meetings |
17 |
|
Average number of members invited to each meeting |
29.2 |
|
Total number of times members were invited to meetings |
496 |
|
Number of recusals |
15 |
|
Number of financial waivers |
1 |
|
Number of appearance authorizations |
7 |
Source: GAO analysis of FDA’s Annual Reports on FDA Advisory Committee Vacancies and Public Disclosures, advisory committee documentation, and FDA officials. | GAO‑26‑107877
Note: FDA’s opioid-related advisory committees are the Anesthetic and Analgesic Drug Products Advisory Committee and the Drug Safety and Risk Management Advisory Committee.
Among the 17 AADPAC and joint AADPAC/DSaRM meetings that we reviewed, FDA granted a waiver to allow one committee member with a financial conflict of interest to participate in the January 2025 AADPAC meeting. This meeting was focused on a biologics license application for an injection to treat leg pain caused by lumbar disc herniation in adults. The committee member owned stock in two competing firms, valued between $25,000 and $50,000, that could have been affected by the committee’s meeting topic. According to the waiver, FDA determined that the member’s expertise was necessary in the interest of public health, and determined that the member’s financial interest was not so substantial as to likely affect the integrity of the member’s expected services. The waiver further stated that FDA considered that the two investments had a cumulative value below 5 percent of the member’s overall non-real estate investments.
In comparison, between June 2018 and the end of fiscal year 2024, the number of financial waivers granted by FDA to members on other CDER advisory committees ranged from one to 24, according to FDA’s annual reports. FDA granted the largest number of waivers—24—during this period to Oncologic Drugs Advisory Committee members. Additionally, FDA granted at least one financial conflict of interest waiver to members on eight other CDER advisory committees. FDA granted zero waivers to members on the nine other CDER advisory committees over this time period. (See appendix II.)
Several researchers have examined the topic of FDA granting waivers to advisory committee members whose expertise is deemed necessary. For example, two of the 16 studies we reviewed focused on the frequency of financial waivers and the most common financial interests among advisory committee members. These studies found that the frequency of waivers granted to members of CDER advisory committees decreased, from 1997 through 2011 (for one study), and from 1997 through 2012 (for the other study).[37] The authors associated the decrease in waivers since late 2008 with the enactment of the Food and Drug Administration Amendments Act of 2007, which capped the number of conflict of interest waivers FDA could grant.[38] (Subsequently, in 2012, the Food and Drug Administration Safety and Innovation Act removed the cap on the number of waivers FDA could grant.[39]) Additionally, one of these studies found that the most common types of reported financial interests among FDA advisory committee members between 1997 and 2011, were investments, as well as consulting activities, research grants, and contracts.[40]
As previously mentioned, FDA reports information on the frequency of financial conflict of interest waivers in the agency’s annual reports on vacancies and public disclosures. For example, in the fiscal year 2024 report, the agency reported that the annual number of waivers, as a percentage of the total number of meeting participants, ranged from zero to almost 4 percent from fiscal years 2015 through 2024.
FDA officials told us that across the 17 AADPAC and joint AADPAC/DSaRM meetings between June 2018 and May 2025, the agency granted seven appearance authorizations for committee members. Officials stated four of these authorizations were for committee members present at AADPAC meetings, while the remaining three were for committee members present at combined AADPAC/DSaRM meetings. In total, according to FDA officials, FDA granted 123 appearance authorizations for all advisory committees across the agency between fiscal years 2018 and 2024. Of these, 87 were granted to CDER committee members. For an earlier time period, researchers also previously examined the frequency with which the agency granted these authorizations across all committees. This study found that FDA granted 201 appearance authorizations across 385 agency-wide advisory committee meetings held between 2008 and 2014.[41]
Once FDA has made its financial conflict of interest waiver determinations, the agency discloses on its website each advisory committee member who received a waiver prior to committee meetings.[42] In our review of the 17 AADPAC and joint AADPAC/DSARM meetings between June 2018 and May 2025, FDA posted the financial waiver it granted to a member for the January 2025 meeting on its website prior to the meeting.[43] Additionally, at each of the 17 AADPAC and joint AADPAC/DSaRM meetings we reviewed, FDA made a public statement about which members, if any, received conflict of interest waivers, consistent with the agency’s guidance.[44] Specifically, at 16 of the meetings, agency officials stated that no conflict of interest waivers were granted. Because FDA granted a financial waiver for the January 2025 meeting, FDA officials verbally disclosed at the start of the meeting which advisory committee member was granted the waiver and why.[45]
Guest Speakers and Open Public Hearing Speakers
We also reviewed the number of financial interest disclosure statements recorded in advisory committee meeting transcripts for invited guest speakers and public speakers during the 17 most recent AADPAC and joint AADPAC/DSaRM meetings. We found that 22 guest speakers presented information at these meetings. Of those 22 guest speakers, 15 made disclosure statements about their financial interests during the committee meetings. Guest speakers disclosed interests such as receiving consulting fees from pharmaceutical companies or serving on scientific advisory boards for organizations whose data were used in FDA-required post-marketing studies. Unlike with advisory committee members, FDA does not report on the number of guest speakers who were invited to meetings and who declined participation or recused themselves from meetings.
FDA does not require members of the public who speak during the open public hearing portion of committee meetings to disclose potential financial interests but encourages them to do so. In our review of the 17 AADPAC and AADPAC/DSaRM meetings between June 2018 and May 2025, we found that of the 137 presentations made by public speakers, 109 presentations included the speaker making a disclosure statement by either stating they had no relevant financial interests or describing their potential conflicts. Of these 109 disclosure statements, most speakers stated that they had no related financial interests. However, several speakers disclosed interests such as having their travel or lodging costs paid for by the drug company whose drug was under review; serving as an expert witness regarding pharmaceutical marketing practices, including the marketing of opioids; being asked to speak at the meeting by the drug company whose drug was under review; or holding current or former roles as an FDA advisory committee chair, drug company consultants or drug company advisors. (See fig. 2.)
Figure 2: Open Public Hearing Speaker Disclosures from the Food and Drug Administration’s (FDA) 17 Most Recent Opioid-Related Committee Meetings from June 2018 to May 2025
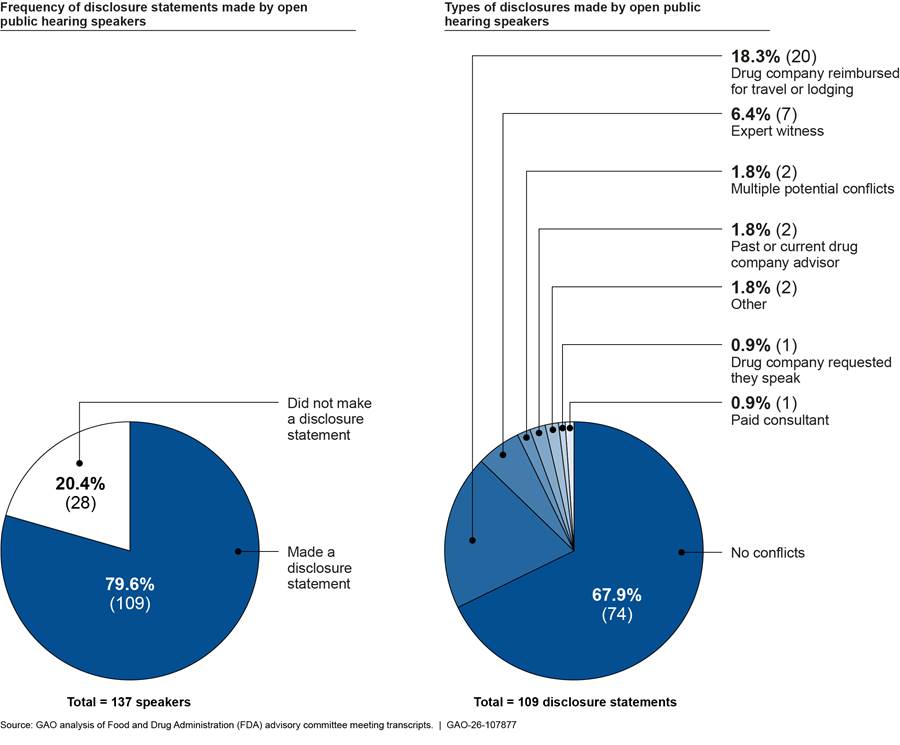
Note: Percentages do not add up to 100 due to rounding. FDA’s opioid-related advisory committees are the Anesthetic and Analgesic Drug Products Advisory Committee and the Drug Safety and Risk Management Advisory Committee. There were 137 open public hearing speakers present across the 17 meetings; however, several speakers may have attended more than one meeting. The “multiple” category includes two open public hearing speakers that disclosed more than one type of potential conflict. One stated they were a former FDA advisory committee chair and had served as an expert witness. Another stated the drug company requested they speak at this meeting and covered their travel expenses. The “other” category includes two open public hearing speakers that disclosed potential conflicts, such as working with national and international organizations or receiving industry funding to develop a product.
While not speaking specifically about opioid committee meetings, six of the 10 stakeholders we spoke with expressed concerns regarding advisory committee open public hearing speakers having financial conflicts of interest. Specifically, several stakeholders expressed concerns about drug companies recruiting patients and compensating representatives to attend and speak at the meetings.
In addition, three studies not specific to opioid committees we reviewed examined the financial disclosures made by open public hearing speakers during meetings of three different FDA advisory committees. These studies found that drug companies seeking approval of their products provide financial incentives to some of the public speakers or their organizations.[46] Further, the three studies found that support provided to speakers often involves travel and lodging provided by the drug company. According to FDA guidance, the open public hearing session of an advisory committee meeting is an opportunity for the public to present relevant information or views. However, FDA staff stated that unlike advisory committee members and guest speakers, they have less control over who is allowed to present information and encourage speakers to disclose conflicts.
FDA Makes Some of Its Advisory Committee Policies Public, But Has Not Shared How It Determines Certain Conflicts or Appearance Issues
FDA publicly shares some of its advisory committee policies, including how the agency determines whether advisory committee members have appearance issues. However, the agency does not make public how it determines whether advisory committee members have financial conflicts of interest or how it determines whether guest speakers have financial conflicts of interest or appearance issues.
FDA Publicly Shares Some of Its Advisory Committee Policies on Its Website
Among the information FDA shares on its advisory committee program website are guidance documents on evaluating appearance issues for advisory committee members, the public availability of committee members’ financial interest information, and the open public hearing portion of advisory committee meetings, during which members of the public can speak.[47] (See fig. 3.) For example, FDA’s guidance on appearance issues for committee members describes examples of circumstances that cause an appearance issue, such as situations when FDA may determine that a member’s interests or relationships more than 1 year old could cause someone to question their impartiality. The guidance also describes some factors the agency may consider when determining if a member should receive an authorization to participate in an advisory committee meeting.
Figure 3: Food and Drug Administration (FDA) Advisory Committee Guidance Documents Website, as of March 2026
FDA’s practice of making guidance documents public is consistent with its own agency-identified best practices. According to the 2024 FDA Report and Plan on Best Practices for Guidance, guidance documents greatly benefit the agency, regulated industry, and the public as a whole by providing consistency and transparency.[48] Posting this information to FDA’s website is also consistent with federal regulations that call for agencies to create public facing websites to help the public understand an agency’s advisory committee program.[49]
In addition to posting guidance documents on the website, FDA posts the federal laws and regulations that the agency follows regarding the standards for convening advisory committees and reviewing potential conflicts of interest. (See fig. 4.)
Figure 4: Food and Drug Administration (FDA) Advisory Committee Laws, Regulations, and Guidance Website, as of March 2026
FDA Has Not Made Public How It Determines Conflicts of Interest or Appearance Issues for Advisory Committees
As of March 2026, FDA’s advisory committee program website did not include information on how it determines whether committee members have financial conflicts of interest, or whether members should participate in meetings. FDA officials told us that prior to 2019, the agency posted guidance to its website on how the agency determined whether committee members have financial conflicts of interest.[50] However, FDA withdrew that guidance in 2019, according to FDA officials, because the agency intended to update its guidance with changes made by the Food and Drug Administration Safety and Innovation Act in 2012 and other agency policy changes.[51] The withdrawn guidance described an algorithm that included 10 steps FDA followed to determine whether an advisory committee member had a financial conflict of interest and if the need for the member’s services outweighed the potential for a conflict created by the financial interest involved. Some stakeholders we spoke to said that FDA’s policies and procedures are not clear to the public, indicating concerns. In addition, some published articles have raised concerns about potential conflicts of interest—such as how advisory committee members who received waivers have voted—and several members of Congress and the public have also raised questions over the years.[52]
The Federal Food, Drug, and Cosmetic Act requires FDA to issue guidance that describes how it reviews the financial interests of committee members that are disclosed to the agency but do not meet the definition of a disqualifying interest under 18 U.S.C. § 208.[53] FDA regulations on good guidance practices generally require the agency to make guidance documents available to the public.[54] According to FDA officials, the agency has been developing new guidance to replace the withdrawn guidance for many years. In July 2025, FDA officials told us that they had drafted this new guidance, but FDA had not yet published the draft guidance for public comment because it remained under review. According to agency officials, FDA expects to make it available for public comment once the review is completed. FDA officials told us the agency will then address the comments and post the final version on its advisory committee guidance website.
However, the guidance development process has continued for many years without a clear time frame for issuing or publicly sharing the guidance. It has been over 6 years since the previous 2008 guidance was withdrawn, and over 13 years since the law requiring FDA to issue the guidance was enacted. Without establishing time frames, it is unclear when FDA will issue or publicly share the guidance, or how much longer the process might take.
Moreover, in the interim period prior to issuing the required guidance, FDA has not shared publicly how the agency determines whether committee members have financial conflicts of interest, or whether members should participate in meetings. Without sharing this information by posting it on the agency’s advisory committee website, FDA leaves the public without knowledge of how the agency makes these determinations, including whether the agency is making them in a reasonable and consistent manner.
Further, FDA has not shared publicly how the agency determines whether guest speakers have financial conflicts of interest, or have appearance issues, or whether they should participate in meetings. For example, while FDA has an internal staff manual that provides a general overview of the disclosure and review process, this information was not posted publicly on FDA’s advisory committee program website as of January 2026. Furthermore, the internal staff manual does not provide details on how FDA determines whether a guest speaker has a conflict of interest. Additionally, the manual does not provide details on how FDA determines whether guest speakers with conflicts or appearance issues should be allowed to participate in committee meetings. Two stakeholders we spoke with said they were unaware that FDA had policies or procedures for determining conflicts of interest or appearance issues for guest speakers. In contrast, for committee members, FDA shares these kinds of details in its guidance on appearance issues. Posting these kinds of details for guest speakers to FDA’s website would provide transparency on how FDA determines whether guest speakers have conflicts of interest or appearance issues.
According to FDA’s 2024 Report and Plan on Best Practices for Guidance, clear, concise, and timely communication through guidance documents is essential to the public health mission of the agency. As previously mentioned, these best practices also state that guidance documents greatly benefit the agency, regulated industry, and the public as a whole by providing consistency and transparency. Providing information, such as through guidance, on how FDA determines committee members’ conflicts of interest and whether they should participate in meetings, as well as information on how FDA determines guest speakers’ financial conflicts of interest and appearance issues, and whether they should participate in meetings would be consistent with these best practices. Providing such information to the public would also be consistent with FDA’s approach of issuing other guidance on advisory committees, as described above.
Sharing information on how FDA determines whether committee members have conflicts of interest and whether guest speakers have conflicts of interest or appearance issues—and whether they should participate in meetings—would also help FDA to address concerns from members of Congress and the public. Sharing this information could also help provide greater assurance that FDA has steps in place to manage conflicts of interest and appearance issues among committee members and guest speakers. In turn, it could help provide a way for members of Congress and the public to better understand FDA’s advisory committee program, ensure accountability and consistency in decision-making, and increase public trust.
Conclusions
FDA advisory committees provide expertise and technical assistance to help the agency make decisions about the development and evaluation of drugs and other products. FDA has worked to build public trust in its advisory committees by posting information on its website about its policies, including how the agency addresses situations when advisory committee members may appear to lack impartiality. In addition, the agency makes public certain results of its conflict of interest review process by posting meeting agendas, transcripts, and information on financial waivers to its website, as shown by our review of advisory committee meetings related to opioids.
However, FDA does not make public how it determines whether advisory committee members have financial conflicts of interest or whether members with conflicts should participate in committee meetings. This is because FDA has yet to finalize the required guidance and has not established a time frame for doing so. Therefore, it is unclear when FDA will issue or publicly share the guidance. FDA has also not posted to its website how it makes these decisions in the interim. Furthermore, FDA does not make public how it determines whether guest speakers have financial conflicts of interest or appearance issues, or whether speakers with conflicts should participate in meetings. Posting information on how FDA determines conflicts of interest and appearance issues, for example in the form of guidance documents, to the agency’s advisory committee program website would help the agency increase transparency, consistent with statements by the FDA Commissioner. It could also provide greater assurance to the public that FDA has steps in place to manage conflicts of interest and to mitigate perceived industry influence on FDA’s decisions.
Recommendations for Executive Action
We are making the following three recommendations to FDA:
The Commissioner of FDA should establish a time frame for issuing and publicly sharing required guidance on how the agency determines financial conflicts of interest for advisory committee members. (Recommendation 1)
The Commissioner of FDA should post to the agency’s advisory committee program website information on how FDA determines whether advisory committee members have financial conflicts of interest and how FDA determines whether they should participate in committee meetings. For example, FDA could post the information it is using to make these determinations while the required guidance is being updated. (Recommendation 2)
The Commissioner of FDA should post to the agency’s advisory committee program website information, such as a guidance document, on how FDA determines whether advisory committee guest speakers have financial conflicts of interest and appearance issues, and how FDA determines whether they should participate in committee meetings. (Recommendation 3)
Agency Comments
We provided a draft of this report to HHS for comment, and its comments are reproduced in appendix III. HHS generally concurred with our recommendations, stating its efforts would include developing guidance and sharing milestones that will be posted to its website.
HHS also provided technical comments, which we incorporated as appropriate.
We are sending copies of this report to the appropriate congressional committees, the Secretary of Health and Human Services, the Commissioner of the Food and Drug Administration, and other interested parties. In addition, the report is available at no charge on the GAO website at http://www.gao.gov.
If you or your staff have any questions about this report, please contact me at DeniganMacauleyM@gao.gov. Contact points for our Offices of Congressional Relations and Media Relations may be found on the last page of this report. GAO staff who made key contributions to this report are listed in appendix IV.

Mary Denigan-Macauley
Director, Health Care
To understand the Food and Drug Administration’s (FDA) advisory committee policies and processes for reviewing and disclosing potential conflicts of interest among committee members, guest speakers, and open public hearing speakers, we interviewed a nongeneralizable selection of 10 researchers and public interest groups (stakeholders). Specifically, we sought an understanding of their experiences participating in or researching FDA advisory committee meetings; and/or their perspectives on FDA’s policies, processes, and management of conflicts of interest among advisory committees.
We identified potential stakeholders to interview from among individuals and organizations with relevant knowledge or perspectives who submitted comments to FDA on advisory committees; published articles on FDA advisory committees; or were referred to us by other stakeholders. From these groups, we selected stakeholders who conducted research on conflicts of interest related to FDA advisory committees, stakeholders with experience working at FDA or participating in FDA meetings by either serving as a member or making presentations to a committee, and stakeholders who expressed a variety of perspectives on FDA’s use of advisory committees.
To select stakeholders, we took the following steps:
· We reviewed the recording of FDA’s public meeting, Optimizing FDA’s Use of Processes for Advisory Committees, held on June 13, 2024, for oral presentations given by individuals regarding conflicts of interest. In addition, we reviewed written public comments submitted by individuals in response to FDA’s public docket for the meeting to identify individuals or organizations that submitted comments related to conflict of interest issues. We interviewed three stakeholders using this selection criteria.
· We identified authors of public policy papers, journal articles, and editorials related to conflicts of interest among FDA advisory committees. We interviewed three stakeholders using this selection criteria.
· We considered suggestions we received from interviewees to identify other individuals or organizations with relevant knowledge or perspectives on FDA advisory committee conflicts of interest. We interviewed three stakeholders using this selection criteria, all of whom also had relevant experience, perspectives or research.
· We selected researchers who led the Initiative on Methods, Measurement, and Pain Assessment in Clinical Trials (IMMPACT) to obtain their perspectives on conflicts of interest that may arise from members participating in their working group and meetings. We interviewed one stakeholder using this selection criteria.
This appendix provides total counts of the number of meetings, number of times members were invited, number of recused members, and number of financial waivers granted among non-opioid Food and Drug Administration (FDA) Center for Drug Evaluation and Research (CDER) advisory committees from June 2018 to the end of fiscal year 2024, according to FDA’s annual reports.
Table 3: Total Counts of Non-Opioid Food and Drug Administration (FDA) Center for Drug Evaluation and Research (CDER) Advisory Committee Meetings, Recusals, and Waivers Between June 2018 and Fiscal Year 2024
|
Advisory committee |
Number of meetings |
Number of times members were invited to meetings |
Number of recusals |
Number of financial waivers granted |
|
Antimicrobial Drugs Advisory Committee |
17 |
416 |
15 |
6 |
|
Arthritis Advisory Committee |
3 |
86 |
7 |
2 |
|
Cardiovascular and Renal Drugs Advisory Committee |
10 |
183 |
7 |
1 |
|
Dermatologic and Ophthalmic Drugs Advisory Committee |
1 |
15 |
2 |
0 |
|
Drug Safety and Risk Management Advisory Committee |
1 |
38 |
0 |
0 |
|
Endocrinologic and Metabolic Drugs Advisory Committee |
8 |
175 |
2 |
0 |
|
Gastrointestinal Drugs Advisory Committee |
4 |
104 |
4 |
6 |
|
Genetic Metabolic Diseases Advisory Committee |
1 |
16 |
0 |
0 |
|
Medical Imaging Drugs Advisory Committee |
2 |
47 |
0 |
0 |
|
Nonprescription Drugs Advisory Committee |
4 |
115 |
1 |
0 |
|
Obstetrics, Reproductive, and Urologic Drugs Advisory Committee |
4 |
128 |
7 |
1 |
|
Oncologic Drugs Advisory Committee |
26 |
673 |
57 |
24 |
|
Peripheral and Central Nervous System Drugs Advisory Committee |
6 |
92 |
7 |
4 |
|
Pharmaceutical Science and Clinical Pharmacology Advisory Committee |
3 |
55 |
2 |
0 |
|
Pharmacy Compounding Advisory Committee |
3 |
82 |
1 |
5 |
|
Psychopharmacologic Drugs Advisory Committee |
8 |
187 |
5 |
2 |
|
Pulmonary-Allergy Advisory Committee |
6 |
154 |
6 |
0 |
|
Total |
107 |
2566 |
123 |
51 |
Source: GAO analysis of FDA’s Annual Reports on FDA Advisory Committee Vacancies and Public Disclosures. | GAO‑26‑107877
Note: The Drug Safety and Risk Management Advisory Committee meetings included in this table are those that were not conducted jointly with the Anesthetic and Analgesic Drug Product Advisory Committee. The counts above do not include counts for meetings that took place in fiscal year 2025, as the FDA annual report on advisory committee vacancies and public disclosures for fiscal year 2025 had not been issued at the time of our review.


GAO Contact
Mary Denigan-Macauley, DeniganMacauleyM@gao.gov.
Staff Acknowledgments
In addition to the contact named above, Will Simerl (Assistant Director), Kendra Sippel-Theodore (Analyst-in-Charge), Sam Amrhein, Sarah Craig, Kaitlin Farquharson, Sarah Mirza, Emily Quick-Cole, Eric Peterson, and Roxanna Sun made key contributions to this report.
The Government Accountability Office, the audit, evaluation, and investigative arm of Congress, exists to support Congress in meeting its constitutional responsibilities and to help improve the performance and accountability of the federal government for the American people. GAO examines the use of public funds; evaluates federal programs and policies; and provides analyses, recommendations, and other assistance to help Congress make informed oversight, policy, and funding decisions. GAO’s commitment to good government is reflected in its core values of accountability, integrity, and reliability.
Obtaining Copies of GAO Reports and Testimony
The fastest and easiest way to obtain copies of GAO documents at no cost is through our website. Each weekday afternoon, GAO posts on its website newly released reports, testimony, and correspondence. You can also subscribe to GAO’s email updates to receive notification of newly posted products.
Order by Phone
The price of each GAO publication reflects GAO’s actual cost of production and distribution and depends on the number of pages in the publication and whether the publication is printed in color or black and white. Pricing and ordering information is posted on GAO’s website, https://www.gao.gov/ordering.htm.
Place orders by calling (202) 512-6000, toll free (866) 801-7077,
or
TDD (202) 512-2537.
Orders may be paid for using American Express, Discover Card, MasterCard, Visa, check, or money order. Call for additional information.
Connect with GAO
Connect with GAO on X,
LinkedIn, Instagram, and YouTube.
Subscribe to our Email Updates. Listen to our Podcasts.
Visit GAO on the web at https://www.gao.gov.
To Report Fraud, Waste, and Abuse in Federal Programs
Contact FraudNet:
Website: https://www.gao.gov/about/what-gao-does/fraudnet
Automated answering system: (800) 424-5454
Media Relations
Sarah Kaczmarek, Managing Director, Media@gao.gov
Congressional Relations
David A. Powner, Acting Managing Director, CongRel@gao.gov
General Inquiries
[1]See 5 U.S.C. § 1002(a).
[2]For this report, we refer to a “potential conflict of interest” when discussing FDA’s review of a member’s financial interests for possible conflicts of interest.
[3]The Federal Advisory Committee Act requires federal agencies to ensure that the advice and recommendations provided by federal advisory committees are independent and transparent to the public. Pub. L. No. 92-463, 86 Stat. 770 (1972) (codified, as amended, at 5 U.S.C. § 1001 et seq.). See also 41 C.F.R. § 102-3.105(j) (requiring agency heads to “[a]ssure that the interests and affiliations of committee members are reviewed for conformance with applicable conflict of interest statutes, regulations issued by the U.S. Office of Government Ethics including any supplemental agency requirements, and other Federal ethics rules”).
[4]See GAO, FDA Advisory Committees: Process for Recruiting Members and Evaluating Potential Conflicts of Interest, GAO‑08‑640, (Washington, D.C.: Sept. 30, 2008).
[5]See GAO, Federal Advisory Committees: Actions Needed to Enhance Decision-Making Transparency and Cost Data Accuracy, GAO‑20‑575, (Washington, D.C.: Sept. 10, 2020).
[6]We recommended that the FDA Commissioner ensure that the Pediatric Advisory Committee renew its charter. In response, the Pediatric Advisory Committee renewed its charter in July 2022 and posted it to the committee’s website and in the General Services Administration’s Federal Advisory Committee Act database. Renewing this charter and posting it online provided greater transparency for Congress and the public, and helped the Pediatric Advisory Committee adhere to its current objectives and obligations. See GAO‑20‑575.
[7]Federal law requires FDA to include industry representatives on certain advisory committees. See, e.g., 21 U.S.C. § 355(n) (requiring FDA to establish advisory committees related to the clinical investigation and approval of new drugs and biologics and include on such committees “a representative of interests of the drug manufacturing industry” not directly affected by matters before the committee). This report does not focus on potential conflicts of interest for industry representatives, due to their specific role representing industry interests.
[8]S. Rep. No. 118-44, at 122-23 (2023).
[9]On March 27, 2025, HHS announced that it would be restructuring the department, including decreasing FDA’s workforce by approximately 3,500 full-time employees, with a focus on streamlining operations and centralizing administrative functions. See HHS, Fact Sheet: HHS’s Transformation to Make America Healthy Again (Washington, D.C.: Mar. 27, 2025). In May 2025, several states challenged HHS’s March 27 announcement on statutory and constitutional grounds. New York v. Kennedy, No. 25-cv-00196 (D.R.I. May 5, 2025). In addition, several other groups challenged government-wide directives requiring federal departments to downsize and reorganize. Am. Fed’n of Gov’t Emps., AFL-CIO v. Trump, No. 25-cv-3698 (N.D. Ca. Apr. 28, 2025). At the time of reporting, both lawsuits were ongoing, and the effects of the proposed restructuring on FDA’s process for determining advisory committee conflicts of interest were yet to be determined.
[10]According to FDA’s website, FDA expert panels are roundtable discussions with independent panels of scientific experts that review the latest scientific evidence, evaluate potential health risks, explore safer alternatives, and offer their recommendations for regulatory action. In 2025, FDA convened five expert panel meetings.
[11]A public health emergency declaration is in effect until the Secretary declares the emergency no longer exists, or 90 days after the declaration, whichever occurs first. A declaration that expires may be renewed by the Secretary. See 42 U.S.C. § 247d(a). Since first being declared a public health emergency in October 2017, the emergency declaration for the opioid crisis has been renewed 33 times, most recently in December 2025.
[12]See, e.g., S. Rep. No. 118-44, at 122-23.
[13]FDA’s website lists the federal laws and regulations that set the standards for convening advisory committees and any guidance FDA has developed describing the agency’s policies on advisory committees.
[14]The following centers and office have advisory committees: Office of the Commissioner, Center for Biologics Evaluation and Research, Center for Devices and Radiological Health, Center for Drug Evaluation and Research, and Center for Tobacco Products.
[15]A committee member may be removed from membership for good cause, which includes a demonstrated bias that interferes with the ability to render objective advice, among other things. 21 C.F.R. § 14.80(e)-(f) (2025).
[16]A special government employee is an officer or employee who is retained, designated, appointed, or employed by the government to perform temporary duties, with or without compensation, not more than 130 days during any period of 365 consecutive days. 18 U.S.C. § 202(a). In general, FDA compensates advisory committee members who are special government employees for their service, reimburses them for travel, and pays them an amount for per diem expenses. 21 C.F.R. § 14.95(a)(1) (2025).
[17]See 21 C.F.R. §§ 14.82 and 14.84 (2025).
[18]21 C.F.R. § 14.80(d) (2025).
[19]See generally 18 U.S.C. § 208 and 5 C.F.R. parts 2635 and 2640 (2025). See also HHS regulations at 45 C.F.R. § 73.735-1003 (2024).
[20]18 U.S.C. § 208(a). See also 5 C.F.R. §§ 2635.401, 2635.402, and 2640.103 (2025).
[21]See 5 C.F.R. § 2635.502 (2025). Ordinarily, FDA’s determination is initiated by information provided to the agency by the committee member, but the agency may make this determination at any time on its own initiative.
[22]The disclosure report asks for information on employment, consultant/advisor roles, contracts/grants/cooperative research, and development agreements that are current or under negotiation. For expert witness and speaking or writing roles, the disclosure report specifies that members should report only on the last 12 months of serving in those roles related to the meeting topic or drug firm. The report does not specify a reporting period for past financial or other relationships. Advisory committee members who are regular government employees may complete a different financial disclosure form—the Office of Government Ethics Confidential Disclosure Report—which requests similar information as the report FDA uses for special government employees, according to FDA officials.
[23]FDA officials told us that they rely on self-disclosure from the members and do not review other potential sources of information because they view other sources as unreliable or not directly relevant.
[24]For the purposes of this report, we use the term “recuse” to describe individuals who were invited but did not participate in a committee meeting because of the potential financial conflict of interest.
[25]18 U.S.C. § 208(b)(1). See also 5 C.F.R. § 2635.402(d)(2) (2025).
[26]18 U.S.C. § 208(b)(3). See also 5 C.F.R. § 2635.402(d)(3) (2025).
[27]5 C.F.R. § 2640.304(a) (2025).
[28]5 C.F.R. § 2635.502(d) (2025).
[29]21 U.S.C. § 379d-1(c)(1)(A).
[30]21 U.S.C. § 379d-1(c)(1)(B).
[31]Specifically, 5 C.F.R. § 2634.901(d) provides that financial disclosure reports and the information they contain are to be withheld from the public. According to FDA, conflict of interest waivers may be released publicly because the agency has a distinct regulatory obligation to release them under 5 C.F.R. § 2640.304(a).
[32]The form FDA uses for guest speakers to disclose financial interests is currently different than the Form 3410 that FDA uses for advisory committee members (FDA’s Confidential Financial Disclosure Report for Special Government Employees). FDA officials said they intend to update the guest speaker financial disclosure form to more closely match the form for advisory committee members. See 88 Fed. Reg. 9,294 (Feb. 13, 2023). In February 2025, FDA officials told us that they expected the revised form to be published in late 2025. However, as of March 2026, the form had not been published.
[33]The meetings we reviewed covered a variety of topics, such as post-marketing requirements for extended-release opioid analgesics, the results of post-marketing studies, various new drug applications, the clinical utility and safety concerns of higher range opioid analgesic dosing, and strategies to increase the availability of naloxone.
[34]FDA does not request information from open public hearing speakers prior to committee meetings; however, the agency does ask speakers to consider disclosing any potential conflicts of interest prior to speaking. See HHS, FDA, Guidance for the Public, FDA Advisory Committee Members, and FDA Staff: The Open Public Hearing at FDA Advisory Committee Meetings (Rockville, MD: May 2013).
[35]Between June 2018 and the end of fiscal year 2024, the Antimicrobial Drugs Advisory Committee recused members 15 times (of 416 invited members), and the Oncologic Drugs Advisory Committee recused members 57 times (of 673 invited members).
[36]In annual reports on advisory committee vacancies and public disclosures, FDA reports the number of persons who were contacted to serve as a member for each advisory committee meeting, but who did not participate because of a potential financial conflict.
[37]Pham-Kanter, Genevieve. “Revisiting Financial Conflicts of Interest in FDA Advisory Committees.” The Milbank Quarterly, vol. 92, No. 3 (2014). Cooper, James C and Golec, Joseph. “Conflicts of Interest on Committees of Experts: The Case of FDA Drug Advisory Committees.” Journal of Law and Economics. (2019).
[38]Pub. L. No. 110-85, § 701, 121 Stat. 823, 900-04. The Food and Drug Administration Amendments Act of 2007 effectively capped the number of waivers by setting a maximum percentage allowable for the total number of meeting exceptions (i.e., waivers) divided by the total number of meeting slots. The allowable percentage decreased by 5 points each fiscal year so that in fiscal year 2008, it was 95 percent and in fiscal year 2012 it was 75 percent.
[39]Pub. L. No. 112-144, § 1142, 126 Stat. 993, 1127-30 (2012).
[40]Pham-Kanter, Genevieve. “Revisiting Financial Conflicts of Interest in FDA Advisory Committees.” The Milbank Quarterly, vol. 92, No. 3 (2014). This study obtained details related to committee members’ financial interests from FDA meeting transcripts, granted financial conflict of interest waivers, and financial conflict of interest waiver acknowledgement forms.
[41]Xu, Jing; Emenanjo, Olubunmi; Ortwerth, Michael; and Lurie, Peter. “Association of Appearance of Conflicts of Interest with Voting Behavior at FDA Advisory Committee Meetings—A Cross-Sectional Study.” JAMA Internal Medicine, vol. 177, No. 7 (2017). Researchers obtained internal agency information on appearance authorizations for all FDA advisory committee meetings held between 2008 and 2014.
[42]As previously mentioned, FDA is not required to publicly disclose information on the appearance authorizations it grants for committee members.
[43]When FDA issues a financial conflict of interest waiver, the agency is required to post the type, nature, and magnitude of the member’s financial interest on its website no later than 15 days in advance of the committee meeting. 21 U.S.C. § 379d-1(c)(1). According to FDA officials, this waiver was posted on December 23, 2024, 18 days prior to the meeting.
[44]See HHS, FDA, Guidance for the Public, FDA Advisory Committee Members, and FDA Staff: Public Availability of Advisory Committee Members’ Financial Interest Information and Waivers (March 2014).
[45]As previously mentioned, FDA officials granted a financial conflict of interest waiver to one committee member for their stock holdings in two competing firms with an aggregate market value between $25,000 and $50,000.
[46]Bertolino, Blake, et al. “Financial conflicts of interest during meetings of cardiovascular and renal drugs advisory committee.” Journal of Osteopathic Medicine. Vol. 122, Issue 9 (2022). This study reviewed open public hearing speakers’ disclosures using FDA meeting transcripts from the Cardiovascular and Renal Drugs Advisory Committee from February 2009 to December 2019.
Roberts, Will, et al. “Characteristics and conflicts of interests of public speakers at Psychopharmacologic Drug and Advisory Committee meetings regarding psychiatric drugs.” BMJ Evidence-Based Medicine. Vol. 25, Issue 4 (2020). This study reviewed open public hearing speakers’ disclosures using FDA meeting transcripts from the Psychopharmacologic Drug Advisory Committee from April 2009 to March 2019.
Abola, Matthew V. and Prasad, Vinay. “Characteristics and Conflicts of Public Speakers at Meetings of the Oncologic Drugs Advisory Committee to the US Food and Drug Administration.” JAMA Internal Medicine. Vol. 176, Issue 3 (2016). This study reviewed open public hearing speakers’ disclosures using FDA meeting transcripts from the Oncologic Drugs Advisory Committee from 2009 to 2014.
[47]HHS, FDA, Draft Guidance for the Public, FDA Advisory Committee Members, and FDA Staff: Procedures for Evaluating Appearance Issues and Granting Authorizations for Participation in FDA Advisory Committees (Silver Spring, Md.: June 2016); HHS, FDA, Guidance for the Public, FDA Advisory Committee Members, and FDA Staff: Public Availability of Advisory Committee Members’ Financial Interest Information and Waivers (Silver Spring, Md.: March 2014); and HHS, FDA, Guidance for the Public, FDA Advisory Committee Members, and FDA Staff: The Open Public Hearing at FDA Advisory Committee Meetings (Silver Spring, Md: May 2013).
[48]See also FDA’s regulations on good guidance practices, specifically 21 C.F.R. § 10.115(g) on the public availability of guidance documents.
[49]See 41 C.F.R. § 102-3.95(b) (2024).
[50]HHS, FDA, Guidance for the Public, FDA Advisory Committee Members, and FDA Staff on Procedures for Determining Conflict of Interest and Eligibility for Participation in FDA Advisory Committees (Silver Spring, Md.: August 2008).
[51]See Pub. L. No. 112-144, § 1142, 126 Stat. at 1127-30. The Food and Drug Administration Safety and Innovation Act removed the statutory cap on the number of conflict of interest waivers FDA could grant.
[52]See, for example, Pham-Kanter, Genevieve. “Revisiting Financial Conflicts of Interest in FDA Advisory Committees.” The Milbank Quarterly, vol. 92, No. 3 (2014). This study reviewed certain FDA advisory committee meetings between 1998 and 2011 and analyzed voting patterns for committee members who received waivers. Among these members, the study found that members with financial ties solely to the companies with drugs under review were more likely to vote in ways favorable to the companies.
[53]21 U.S.C. § 379d-1(g). This requirement was added in 2012 with the enactment of the Food and Drug Administration Safety and Innovation Act. Pub. L. No. 112-144, § 1142(a)(5), 126 Stat. at 1130.
[54]21 C.F.R. § 10.115(g) (2025).
