MEDICARE PART D
Implementation of Beneficiary Premium Stabilization Demonstration
Report to Congressional Requesters
United States Government Accountability Office
A report to congressional requesters.
For more information, contact: John E. Dicken at dickenj@gao.gov.
What GAO Found
The Centers for Medicare & Medicaid Services (CMS)
implemented the voluntary Medicare Part D Premium Stabilization Demonstration
(Demonstration) in 2025 to stabilize beneficiary monthly premiums and
enrollment in Part D standalone prescription drug plans. Nearly all plan sponsors
opted to participate. Without the Demonstration, GAO’s analysis of CMS data
showed that, if beneficiaries in standalone drug plans in 2024 remained in
their plan in 2025, their monthly premium would have nearly doubled, on
average. In addition, monthly premiums for 37 percent of these beneficiaries
would have increased by more than $40 (see figure). If these premium increases
had taken effect, CMS officials expected widespread changes in enrollment for
beneficiaries in standalone drug plans, which could disrupt beneficiaries’ access
to their medications.
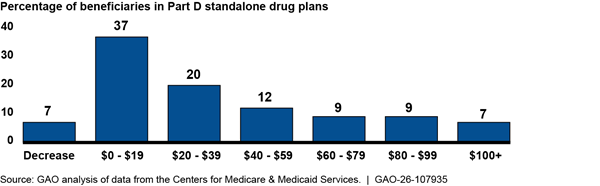
Notes: Results indicate how premiums could have changed if beneficiaries in standalone Part D plans in 2024 had remained in the same plan or were transferred to another one in 2025. Results are weighted based on 2024 enrollment of beneficiaries who were not eligible for the low-income subsidy.
To stabilize premiums with the goal of stabilizing enrollment in standalone drug plans, CMS (1) reduced beneficiary premiums in 2025 by up to $15 and then (2) limited each plan’s premium increases to $35 from 2024 to 2025. CMS also provided additional protection for plan sponsors in 2025. For 2026, CMS provides smaller premium reductions and allows for greater premium increases. Collectively, CMS officials estimated that the Demonstration would cost a total of $9.8 billion in 2025 and 2026.
The Department of Health and Human Services’ Office of the Assistant Secretary for Planning and Evaluation (ASPE), through an agreement with CMS, designed an evaluation framework to be used to determine whether the Demonstration achieved its goals. GAO’s analysis of CMS data showed that average premiums in standalone drug plans increased from $42 in 2024 to $43 in 2025 under the Demonstration for beneficiaries not eligible for the low-income subsidy. In addition, enrollment in these plans increased by 2 percent from 2024 to 2025, while the percentage of all Part D enrollees in standalone plans remained at 42 percent. However, an evaluation is necessary to determine the extent to which changes such as these were due to the Demonstration and not to other factors, such as changes to the Part D drug benefit. ASPE officials told GAO that they plan to continue their evaluation efforts in fiscal year 2026. GAO received a copy of the finalized evaluation framework in January 2026.
Why GAO Did This Study
The Medicare Part D program provides voluntary outpatient prescription drug coverage to beneficiaries, including those enrolled in standalone drug plans. The Inflation Reduction Act of 2022 required significant changes to the Part D drug benefit, some of which took effect in 2025.
CMS reported in July 2024 that increased variation in plan sponsors’ expected costs for providing drug coverage in 2025 could lead to substantial premium increases for beneficiaries. CMS subsequently announced in July 2024 the Demonstration for standalone drug plans, using its authority under section 402 of the Social Security Amendments of 1967 as amended. GAO was asked to review the legality of the Demonstration and issued a legal decision in May 2025 concluding that this Demonstration, as implemented for 2025, was consistent with this statutory authority.
GAO was also asked to review other aspects of the Demonstration. This report describes CMS’s (1) implementation of the Demonstration, and (2) plan to evaluate the Demonstration.
GAO reviewed CMS documents, such as those about the development and evaluation of the Demonstration, and CMS data on premiums for Part D standalone drug plans. In its analysis of standalone drug plan premiums, GAO focused on beneficiaries who were not eligible for the low-income subsidy and, therefore, were required to pay plan premiums. GAO also interviewed CMS and ASPE officials, Part D plan sponsors, and organizations representing Medicare beneficiaries.
|
Abbreviations |
|
|
ASPE |
The Office of the Assistant Secretary for Planning and Evaluation |
|
CMS |
Centers for Medicare & Medicaid Services |
|
HHS |
Department of Health and Human Services |
|
IRA |
Inflation Reduction Act of 2022 |
|
MA |
Medicare Advantage |
This is a work of the U.S. government and is not subject to copyright protection in the United States. The published product may be reproduced and distributed in its entirety without further permission from GAO. However, because this work may contain copyrighted images or other material, permission from the copyright holder may be necessary if you wish to reproduce this material separately.
February 26, 2026
The Honorable Mike Crapo
Chairman
Committee on Finance
United States Senate
The Honorable Brett Guthrie
Chairman
Committee on Energy and Commerce
House of Representatives
The Honorable Jason Smith
Chairman
Committee on Ways and Means
House of Representatives
The Medicare Part D program provided voluntary outpatient prescription drug coverage to approximately 55 million beneficiaries in 2025. Of those, 42 percent—or approximately 23 million beneficiaries—were enrolled in standalone Part D prescription drug plans (standalone drug plans) offered by Part D plan sponsors and 58 percent were enrolled in Medicare Advantage (MA) plans that offer drug coverage (MA drug plans).
The Inflation Reduction Act of 2022 (IRA) included a provision limiting increases in Part D premiums—the monthly fee Medicare beneficiaries pay for prescription drug coverage.[1] However, the Centers for Medicare & Medicaid Services (CMS) reported in July 2024 that plan sponsors’ expected average costs (called plan bids) for providing prescription drug coverage to beneficiaries had increased 179 percent from 2024 to 2025 and could lead to substantial premium increases for beneficiaries.[2]
CMS determined that the IRA’s limit on beneficiaries’ base premium increases, on its own, was not enough to mitigate the overall premium increases that would have occurred in 2025.[3] As such, also in July 2024, CMS announced the voluntary Medicare Part D Premium Stabilization Demonstration (Demonstration) for standalone drug plans. The agency planned for the Demonstration to last for at least 3 years—2025, 2026, and 2027. Nearly all standalone drug plans opted to participate in this Demonstration in 2025. According to CMS, the agency used its authority under section 402 of the Social Security Amendments of 1967, as amended, to implement the Demonstration, which started in January 2025.[4] We were asked to review the legality of the Demonstration and issued a legal decision in May 2025 concluding that this Demonstration, as implemented for 2025, was consistent with the Secretary’s statutory authority under Section 402.[5]
You asked us to review the Medicare Part D Premium Stabilization Demonstration. This report describes (1) CMS’s implementation of the Part D Demonstration, and (2) CMS’s plan to evaluate the Part D Demonstration.
To describe CMS’s implementation of the Demonstration, we reviewed relevant CMS documents, including those about the development of the Demonstration and those estimating its budgetary effects. Further, we used CMS data on Part D standalone drug plans bids and premiums to compare premiums in 2024 to (1) what premiums would have been in 2025 absent the Demonstration if beneficiaries had remained in their plan in 2025, and (2) 2025 premiums under the Demonstration.[6] We weighted our results using beneficiary enrollment in standalone drug plans in 2024 and 2025, as appropriate.[7] To assess the reliability of the Part D bid and premium data, we reviewed relevant documentation and interviewed CMS officials. We found these data to be sufficiently reliable for the purposes of our analysis of standalone drug plan premiums in 2024 and 2025. Additionally, we interviewed CMS officials, or received written responses, about their implementation of the Demonstration, including about the underlying data used to inform their decision making and their plans for the Demonstration in subsequent years. We also interviewed representatives of two selected organizations representing Medicare beneficiaries and five selected large Part D plan sponsors to obtain their perspectives of their experiences with the Demonstration.[8] The views of these organizations and plan sponsors are not generalizable.
To describe CMS’s plan to evaluate the Demonstration, we reviewed relevant documents about the plan to develop an evaluation design, such as those between CMS and the Department of Health and Human Services’ Office of the Assistant Secretary for Planning and Evaluation (ASPE); and from ASPE’s contractor. ASPE and its contractor are responsible for designing the evaluation. These include the Statement of Work for designing the evaluation and the interagency agreement between CMS and ASPE that assigns responsibility to the latter for developing the evaluation design. Additionally, we interviewed officials from CMS and ASPE about their plans to evaluate the Demonstration, including when the evaluation design will be completed and any challenges with designing and ultimately conducting the evaluation. We also asked officials about how they plan to use the findings from the evaluation, once completed.
We conducted this performance audit from November 2024 to February 2026 in accordance with generally accepted government auditing standards. Those standards require that we plan and perform the audit to obtain sufficient, appropriate evidence to provide a reasonable basis for our findings and conclusions based on our audit objectives. We believe that the evidence obtained provides a reasonable basis for our findings and conclusions based on our audit objectives.
Background
CMS, the agency within the Department of Health and Human Services (HHS) responsible for overseeing the Medicare program, provides outpatient prescription drug coverage to Medicare beneficiaries through drug plans provided by Part D plan sponsors—private companies that contract with CMS to provide this drug coverage.[9]
Part D Drug Benefit
Medicare beneficiaries receive coverage for the Part D drug benefit through either of the following.
· Standalone prescription drug plans that supplement traditional Medicare with prescription drug coverage.
· MA plans, which generally must cover all traditional Medicare benefits, that opt to include Part D prescription drug coverage.[10]
Both standalone and MA drug plans that offer Part D coverage must offer a minimum “standard benefit” package by meeting certain statutory requirements. There are three different Part D coverage phases in the standard benefit, in which the beneficiary, the plan sponsor, the drug manufacturer, and Medicare program pay different amounts.[11]
· Deductible phase. In the deductible phase, beneficiaries pay all out-of-pocket prescription drug costs until they reach a specific deductible amount, or threshold, before the plan sponsor pays for coverage.[12] In 2025, the standard benefit had a deductible phase where the beneficiary was responsible for 100 percent of all drug costs, up to $590.
· Initial coverage phase. Beneficiaries enter the initial coverage phase once a beneficiary meets the deductible amount. In the initial coverage phase, beneficiaries are responsible for 25 percent of prescription drug spending until they reach the total out-of-pocket limit, which was $2,000 in 2025.
· Catastrophic coverage phase. After reaching the out-of-pocket limit, beneficiaries enter the catastrophic coverage phase in which beneficiaries are not responsible for prescription drug costs for covered Part D drugs. In the catastrophic coverage phase, the plan sponsor, Medicare, and drug manufacturers cover all prescription drug costs. (See fig. 1).
Figure 1: Share of Part D Brand-Name Prescription Drug Costs in Medicare Part D Standard Benefit in 2025, by Coverage Phase
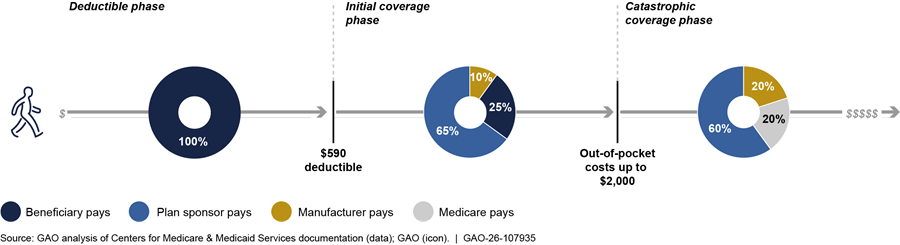
Note: This figure describes the phases of the Medicare Part D standard drug benefit for beneficiaries not receiving low-income subsidy assistance, including the amounts spent by plan sponsors, beneficiaries, manufacturers, and Medicare on applicable, brand-name prescription drugs. Part D plan sponsors are required to offer a standard benefit but may provide alternative benefit structures that are actuarially equivalent to the standard benefit. The standard benefit includes different coverage phases with costs split between the beneficiary, plan, drug manufacturer and Medicare. In 2025, beneficiaries paid 100 percent of drug costs in the deductible phase, up to $590, and out-of-pocket costs were capped out $2,000.
Plan sponsors may follow this standard benefit structure in their plans, or they may offer alternative benefit structures. That is, they may offer a benefit that is actuarially equivalent to the standard benefit structure subject to some constraints but adjust certain cost-sharing features, while covering the same share of drug spending for beneficiaries. In addition to the standard benefit, some plans offer supplemental coverage for which beneficiaries pay an additional premium. Those plans are sometimes referred to as enhanced plans. Benefits offered by enhanced plans may lower beneficiary cost-sharing, offer a lower deductible, or offer supplemental drug coverage.
Medicare Part D Payments to Plan Sponsor and Beneficiary Premiums
Payments to plan sponsors. Medicare makes two primary payments to plan sponsors, as follows.
· Prospective monthly payments. Medicare pays plan sponsors direct subsidies in the form of a monthly capitated payment to cover the estimated costs of providing beneficiary drug coverage. These payments are determined through annual bids submitted by plan sponsors in June preceding the program year, which runs from January 1 through December 31. Those bids reflect the plan sponsors’ estimates of expected costs for a Medicare beneficiary of average risk that the sponsor expects to incur during the ensuing program year. The monthly payment for a specific beneficiary puts plan sponsors at financial risk because they incur losses if actual drug costs exceed the payment. However, they make profits if a beneficiary’s actual drug costs are lower than the payment.[13]
· Reinsurance. Medicare pays plan sponsors for a share of a beneficiary’s prescription drug costs in the catastrophic phase of the drug benefit.
Additionally, Medicare Part D risk sharing arrangements limit a plan sponsor’s overall losses or profits. Under these risk sharing arrangements (known as “risk corridors”), Medicare shares with plan sponsors any high costs or recoups part of any excessive gains when a plan has costs above or below its target amount, respectively. For example, after the benefit year ends, if the actual costs incurred by the plan sponsor for all Medicare beneficiaries are higher than estimated in its bid, Medicare pays a portion of the excess costs above a certain threshold. On the other hand, if actual costs incurred by the plan sponsor for all Medicare beneficiaries are lower than estimated costs in the bid, the plan pays a portion of the savings back to the Medicare program. (See fig. 2.) Medicare makes risk corridor payments to plan sponsors after the benefit year ends.
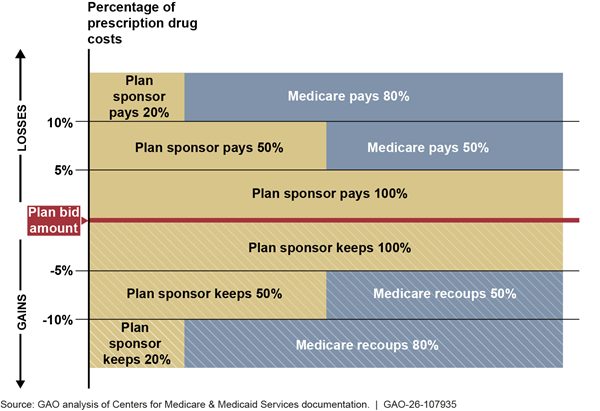
Note: Medicare Part D risk corridors are risk sharing arrangements that limit excessive loss or gains for each Part D plan sponsor when actual costs are above or below a plan sponsor’s bid amount. Part D plan sponsor bids reflect the expected costs of providing the Part D benefit to a beneficiary with average prescription drug costs. Under risk corridors, Medicare limits a plan sponsor’s potential losses (or gains) by financing some of the higher-than-expected costs (or by recouping excessive profits). Medicare makes risk corridor payments to plan sponsors after the benefit year ends. For example, after the benefit year ends, if the actual costs incurred by the plan sponsor for all Medicare beneficiaries are higher than estimated in its bid, Medicare pays a portion of the excess costs above a certain threshold. Alternatively, if actual costs incurred by the plan sponsor for all Medicare beneficiaries are lower than estimated costs in the bid, the plan pays a portion of the savings back to the Medicare program. Figure reflects current law thresholds for 2025. See 42 U.S.C. § 1395w-115(e); 42 C.F.R. § 423.336 (2025).
Beneficiary premiums. In addition to a deductible and cost sharing, most plan sponsors charge beneficiaries monthly premiums for their drug coverage, which vary by plan. Beneficiary premiums are determined in part by the base beneficiary premium, which is calculated as a percentage of the nationwide average of bids for standard benefits submitted to CMS by plan sponsors.[14] In general, the actual beneficiary premium depends on how the plan sponsor’s bid compares to the nationwide average bid. For example, when a plan sponsor’s bid is higher than the national average, beneficiaries pay the base premium plus the amount by which the plan’s bid exceeds the national average. In 2025, the base beneficiary premium of $37 increased by about 6 percent, or about $2, relative to $35 in 2024. This reflected CMS’s calculations based on the nationwide average plan bid as well as the IRA requirement to limit annual base beneficiary premium increases to 6 percent.[15] Without this 6 percent limit, base beneficiary premiums would have been increased by 61 percent from $35 in 2024 to $56 in 2025.[16]
CMS also provides subsidies for beneficiaries who qualify for low-income assistance to help cover certain out-of-pocket costs (e.g., deductibles or copayments). In 2025, about 13 million Part D beneficiaries (about 24 percent) received low-income subsidies, including 4 million beneficiaries (about 33 percent of low-income beneficiaries) in standalone drug plans and 9 million beneficiaries (about 67 percent of low-income beneficiaries) in MA drug plans.[17]
The Inflation Reduction Act of 2022
The IRA made significant changes to Medicare’s prescription drug benefit, which began taking effect in 2023 with certain provisions taking effect in later years.[18] For example, in 2025, the following Part D benefit changes took effect.
· Capped beneficiaries’ total Medicare Part D out-of-pocket spending at $2,000 compared to about $8,000 in 2024.[19]
· Increased plan sponsors’ share of prescription drug costs after a beneficiary reaches the out-of-pocket spending cap (in the catastrophic phase), from 20 percent in 2024 to 60 percent in 2025 for all covered drugs.
· Reduced Medicare’s share of cost in the catastrophic phase from 80 percent in 2024 to 20 percent in 2025 for brand drugs and 40 percent for generic drugs.
Section 402 Demonstration Authority
Under Section 402 of the Social Security Amendments of 1967, as amended, the Secretary of Health and Human Services (Secretary) has authority to develop and implement projects, called demonstrations, to test certain changes in Medicare payment methods. This authority allows the Secretary to waive compliance with certain Medicare payment and reimbursement policies to conduct these demonstrations. Specifically, Section 402 gives the Secretary authority to engage in demonstration projects “to determine whether, and if so, which, changes in methods of payment or reimbursement…would have the effect of increasing the efficiency and economy of health services…through the creation of additional incentives to these ends without adversely affecting the quality of such services” under Medicare.[20]
Over the years, CMS has conducted several demonstrations using this authority to test policies that might address transitional issues associated with the implementation of major changes to the Medicare program. As of March 2025, CMS officials identified nine demonstrations the agency had conducted under its Section 402 authority since 1995.[21] Four of these demonstrations sought, or are currently seeking, to test approaches to limit fluctuations in beneficiary premiums in Medicare Part D prescription drug benefit programs. The three completed demonstrations varied in costs, from being designed as budget neutral to costing $778 and $887 million. (See app. I for more detail on these and other Medicare demonstrations conducted under Section 402 authority.)
The current Demonstration under Section 402 authority, the Medicare Part D Premium Stabilization Demonstration, was open to all standalone Part D plan sponsors offering Part D prescription drug plans.[22] In 2025, 39 plan sponsors offered 818 standalone drug plans; of these, plan sponsors for 782 plans (96 percent) opted to participate in the first year of the voluntary Demonstration. According to CMS guidance in 2024, only plan sponsors that participated in the Demonstration in 2025 could opt to participate in future years of the Demonstration.
CMS Implemented the Part D Demonstration to Determine Whether Stabilizing Premiums Would Stabilize Enrollment
CMS Determined That Potential Premium Increases in 2025 Could Result in Widespread Enrollment Changes
According to CMS officials, the agency implemented its Part D Premium Stabilization Demonstration because premiums in standalone drug plans were going to increase for many beneficiaries in 2025 and could result in widespread enrollment changes in these plans.
Potential premium increases. According to CMS officials, premiums in standalone drug plans were going to increase for many beneficiaries in 2025. Our analysis of CMS data confirms that without the Demonstration, premiums for standalone plan beneficiaries not eligible for the low-income subsidy could have nearly doubled if they had remained in their plan in 2025. Specifically, our analysis of CMS data showed that, based on the bids CMS received, the average monthly premium for these beneficiaries was projected to increase from approximately $43 in 2024 to $81 in 2025, if they had remained in the same plan in 2025.[23]
Further, if these standalone drug plan beneficiaries in 2024 had remained in their same plans in 2025, premiums for an estimated 3.9 million beneficiaries (30 percent) of them would have increased by between $40 and $100 per month absent the Demonstration, according to our analysis of CMS data. Additionally, premiums for nearly 1 million of them (7 percent) would have increased by $100 or more per month. See table 1 for more detail.
Table 1: Potential Monthly Part D Standalone Plan Premium Increases for Beneficiaries from 2024 to 2025, Absent CMS’s Part D Premium Stabilization Demonstration
|
|
Beneficiaries in Standalone Drug Plans |
|
|
Total proposed 2025 premium compared to total 2024 premium |
Number |
Percent |
|
Decreasing |
861,971 |
7% |
|
No change |
952 |
0% |
|
Increasing to less than $10 |
762,863 |
6% |
|
Increasing $10 to less than $20 |
4,132,652 |
31% |
|
Increasing $20 to less than $30 |
889,454 |
7% |
|
Increasing $30 to less than $40 |
1,676,962 |
13% |
|
Increasing $40 to less than $50 |
894,817 |
7% |
|
Increasing $50 to less than $60 |
687,612 |
5% |
|
Increasing $60 to less than $70 |
705,761 |
5% |
|
Increasing $70 to less than $80 |
472,121 |
4% |
|
Increasing $80 to less than $90 |
355,290 |
3% |
|
Increasing $90 to less than $100 |
752,553 |
6% |
|
Increasing $100+ |
975,183 |
7% |
Source: GAO analysis of Centers for Medicare & Medicaid Services (CMS) data. | GAO‑26‑107935
Note: This table displays calculations of potential premium increases for beneficiaries enrolled in standalone Medicare Part D prescription drug plans that existed in both 2024 and 2025 (including beneficiaries who would have been transferred to another plan) and reflects the Inflation Reduction Act of 2022’s’s requirement to limit annual base beneficiary premium increases to 6 percent. See 42 U.S.C. § 1395w-113(a)(8). Further, the analyses are based on weighted July 2024 enrollment of non-low-income subsidy beneficiaries. We limited our analysis of CMS data to non-low-income subsidy beneficiaries because these beneficiaries do not receive the low-income subsidy. Therefore, non-low-income subsidy beneficiaries pay Part D premiums determined by CMS and are most affected by annual premium increases. Comparatively, beneficiaries eligible for the low-income subsidy can enroll in certain plans without paying a premium.
Percentages may exceed 100 percent due to rounding.
Reasons for potential premium increases. CMS officials largely attributed the potential premium increases for standalone drug plans to the Part D benefit design changes that went into effect in 2025, which increased financial liability and uncertainty for plan sponsors. CMS cited the following examples of Part D benefit changes that increased plan sponsors’ financial liability:
· Increased plan sponsor financial liability in catastrophic phase of the Part D benefit. Relative to 2024, plan sponsor liability in the catastrophic phase increased in 2025 from 20 percent to 60 percent of gross Part D costs (for brand-name and generic drugs). This shift in liability from Medicare to plan sponsors—resulting from decreasing reinsurance payments from Medicare and increasing direct subsidies—puts plan sponsors at greater financial risk.[24]
· Lower cap on beneficiary out-of-pocket costs. CMS cited the lowering of this cap to $2,000 in 2025 as another reason for the premium increase because plan sponsors would be largely liable for remaining expenses for beneficiaries whose out-of-pocket costs exceed the cap. Although lower cost sharing for beneficiaries at the point of sale may make medications more affordable, it is also likely to put upward pressure on overall drug utilization and benefit costs and introduce more uncertainty for plan sponsors.[25] For example, because beneficiaries who reach the out-of-pocket cap are no longer responsible for their drug costs, they may increasingly use specialty and high-cost drugs.[26] Increased utilization of these drugs by beneficiaries could add to plan sponsors’ costs for covering the drugs and possibly to increased premiums. Further, researchers have emphasized that the expected growth in utilization and costs of Part D-covered drugs are critical assumptions plan sponsors must account for in their annual bid submissions.[27]
CMS officials confirmed that the new Part D benefit changes informed plan sponsors’ bids for 2025, contributing to variation in the bids and to potential premium increases for some beneficiaries.[28] We observed greater variation in standalone drug plan bids in 2025 than in 2024 in our review of CMS’s data. For example, we found that the percentage of beneficiaries in standalone drug plans with bids that exceeded the nationwide average by more than $50 increased from 6 percent in 2024 to 28 percent in 2025.[29] This increased variation contributes to potential premium increases because, in general, a plan’s premium for basic benefits is the sum of the base beneficiary premium and the difference between the plan’s bid and the nationwide average bid (see sidebar).[30] Plan sponsors we spoke with also confirmed that their increased financial liability in the catastrophic phase and the lower out-of-pocket costs for beneficiaries helped inform their bids, and therefore affected premiums, for 2025.
|
Calculating Medicare Part D Premiums for Beneficiaries Medicare Part D beneficiary premiums are determined in part by the base beneficiary premium, which is calculated as a percentage of the nationwide average of bids (or expected average costs for providing prescription drug coverage), submitted by plan sponsors to the Centers for Medicare & Medicaid Services for standard Part D benefits. In general, the actual beneficiary premium for a Part D plan depends on how the plan sponsor’s bid compares to the nationwide average bid. For example: · When a plan sponsor’s bid is equal to the national average, the beneficiary premium is equal to the base beneficiary premium. In 2025, the base beneficiary premium amount was about $37, and in this situation the beneficiary premium would equal about $37. · When the plan sponsor’s bid is lower than the national average, beneficiaries pay the base premium minus the amount by which the plan’s bid is below that of the national average. For example, if a plan’s bid is $5 lower than the national average, the beneficiary premium would be $37 minus $5, or $32, in 2025. · When a plan sponsor’s bid is higher than the national average, beneficiaries pay the base premium plus the amount by which the plan’s bid exceeds the national average. For example, if a plan’s bid is $20 higher than the national average, the beneficiary premium would be $37 plus $20, or $57, in 2025. Source: GAO analysis of documentation from the Centers for Medicare & Medicaid Services and MedPAC | GAO‑26‑107935. |
Implications of potential premium increases for Part D enrollment and for beneficiaries. According to CMS officials, the potential monthly premium increases described above could have had implications for the Part D market for standalone drug plans to the extent they led beneficiaries in these plans to change their Part D enrollment—such as by switching from standalone drug plans to MA plans or changing standalone plans. CMS officials also explained how changing enrollment can potentially impact beneficiaries’ access to care and drug utilization.
Potential effects on enrollment in standalone drug plans. Enrollment in standalone drug plans has declined in recent years. For example, enrollment in standalone drug plans declined by an average of about 2 percent per year from 25.5 million beneficiaries in 2018 to 23.0 million beneficiaries in 2024. Further, the share of Part D enrollment in MA drug plans has increased, consistent with the overall shift in enrollment from traditional Medicare to MA plans (see sidebar).[31] The potential premium increases in 2025 for standalone plans described above would have widened the gap between the Part D premiums of standalone and MA drug plans, thus potentially motivating more beneficiaries to make the switch from standalone to MA drug plans.[32]
|
Shifting Enrollment in Medicare Part D Plans Part D enrollment has increasingly shifted from standalone drug plans to Medicare Advantage (MA) drug plans in recent years. For instance, in 2018, 41 percent (about 18 million) of Medicare Part D beneficiaries were enrolled in MA drug plans. By 2025, that number had increased to 58 percent (about 32 million). This shift in enrollment was accompanied by other changes in the market for standalone drug plans, as described below. Fewer standalone plan options. In 2025, beneficiaries had fewer options to select from for prescription drug coverage. While the availability of options varied across different geographic areas, beneficiaries could, on average, choose from 14 distinct standalone drug plan coverage options (seven fewer than in 2024), compared to 34 different options for drug coverage as part of an MA plan. The number of standalone benchmark plans—those offered without premiums to beneficiaries receiving low-income subsidies—has also declined over the years. In 2025, on average, there were four benchmark plans per region, down from 10 in 2014. Additionally, five of the 34 standalone drug plan regions only had two benchmark plan offerings in 2025. Greater concentration in standalone plan market. The percentage of total enrollment in standalone drug plans accounted for by the five largest plan sponsors increased from 83 percent in 2018 to 91 percent in 2025. Source: GAO analysis of documentation from the Centers for Medicare & Medicaid Services, KFF, and MedPAC | GAO‑26‑107935. |
Because of the potential premium increases that could occur absent the Demonstration, CMS officials said they expected to see higher rates of beneficiaries changing their Part D enrollment in 2025.[33] Plan sponsors also projected sizable reductions in their standalone drug plans. Specifically, CMS officials told us they calculated that, based on their review of the plan sponsors’ bids, plan sponsors projected an enrollment decrease of 27 percent for their drug plans, on average, from 2024 to 2025 absent the Demonstration. This expected 27 percent reduction would have far exceeded the 2 percent annual decline described above from 2018 through 2024.[34] CMS officials also noted that this expected reduction in standalone drug plan enrollment could accelerate the shift in enrollment from standalone to MA drug plans, whose Part D premiums were significantly lower, after accounting for premium reductions due to MA rebates.[35]
Potential effects on beneficiaries. The anticipated monthly premium increases could also have implications for beneficiaries, according to CMS officials.[36] For instance, an increased premium may motivate a beneficiary to change to a new plan with a lower premium. However, changing to a new plan could disrupt their access to medication if the new plan has higher out of pocket payments for their current medications, or does not cover a specific medication.[37] Staff from Medicare beneficiary advocacy groups we spoke with echoed these concerns about loss of access to essential medications due to premium increases in 2025.
CMS Implemented the Demonstration to Stabilize Enrollment in, and Bids for, Standalone Part D Plans at an Estimated Cost of $9.8 Billion in 2025 and 2026
CMS implemented the voluntary Medicare Part D Premium Stabilization Demonstration in 2025 for at least 3 years to achieve two goals at an estimated cost of approximately $9.8 billion for 2025 and 2026, according to CMS officials.[38] In 2025, 782 of 818 standalone drug plans participated.
Demonstration Goals
CMS officials told us they generally had two goals for the Demonstration: (1) to stabilize beneficiary enrollment in standalone drug plans by stabilizing beneficiary premiums, and (2) to stabilize bids for standalone drug plans in future years by providing temporary, additional financial protection for plan sponsors.
Stabilizing enrollment. To meet the first goal of stabilizing enrollment in standalone drug plans, CMS adjusted premiums for beneficiaries in two ways in 2025.
· Reduced the base beneficiary premium for all participating plans—and therefore all beneficiaries—by up to $15 in 2025.[39] This reduction diminished the gap between the Part D premiums of standalone drug plans and MA drug plans.
· Established a $35 limit on premium increases from 2024 to 2025 to address any high premium increases that remained after the $15 reduction.[40]
Overall, the average monthly premium for standalone drug plan beneficiaries not eligible for the low-income subsidy increased from $42 in 2024 to $43 in 2025 under the Demonstration, according to our analysis of CMS data.[41] In addition, enrollment in standalone drug plans increased by approximately 2 percent from 23.0 million in 2024 to 23.4 million in 2025.
Stabilizing future bids for standalone drug plans. CMS officials’ stated second goal of the Demonstration was to provide additional financial protection for plan sponsors to stabilize their bids in future years. This temporary financial protection allows plans to gain experience with the effects of the Part D benefit changes required by the IRA, which could help them stabilize, or submit more accurate, bids in the future, according to CMS officials.[42] To do so, CMS modified the Part D risk corridors for standalone drug plans to have Medicare assume a greater portion of potential financial losses that participating plan sponsors incur if their actual costs of providing Part D benefits exceed what they expected. For example, absent the Demonstration, Medicare would have begun sharing a plan sponsor’s unanticipated financial losses when those losses exceeded 5 percent of expected costs. Under the Demonstration, CMS reduced this threshold to 2.5 percent of a plan sponsor’s unanticipated losses. However, no corresponding change was made to the risk corridors for unexpected gains. (See fig. 3.)
Figure 3: Medicare Part D Risk Corridors Under the First Year (2025) of the Premium Stabilization Demonstration for Participating Plan Sponsors
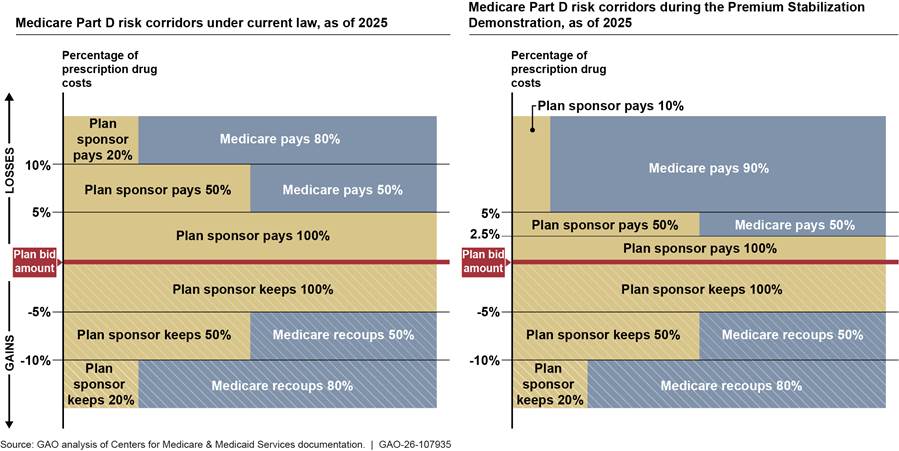
Note: Medicare Part D risk corridors are risk sharing arrangements that limit excessive losses or gains for each Part D plan sponsor when actual costs are above or below a plan sponsor’s bid amount. Under risk corridors, Medicare limits a plan sponsor’s potential losses (or gains) by financing some of the higher-than-expected costs (or by recouping excessive profits). Medicare makes risk corridor payments to plan sponsors after the benefit year ends. For example, after the benefit year ends, if the actual costs incurred by the plan sponsor for all Medicare beneficiaries are higher than estimated in its bid, Medicare pays a portion of the excess costs above a certain threshold. Alternatively, if actual costs incurred by the plan sponsor for all Medicare beneficiaries are lower than estimated costs in the bid, the plan pays a portion of the savings back to the Medicare program. The Centers for Medicare & Medicaid Services modified the risk corridors for plan sponsors participating in the Part D Premium Stabilization Demonstration in 2025.
Some of the plan sponsors’ reasons for participating in the Demonstration echoed the goals cited by CMS. Specifically, all five plan sponsors in our review told us they decided to participate in the Demonstration to help protect beneficiaries from significant premium increases that could have led to enrollment changes. In addition, representatives we interviewed from two of the five plan sponsors said they participated in the Demonstration in part because the government’s greater risk-sharing for potential losses under the revised risk corridors helped provide some stability.
Demonstration Costs
CMS has estimated the Demonstration would cost a total of $9.8 billion in 2025 and 2026, including $6.2 billion in 2025 and $3.6 billion in 2026.
Estimated 2025 costs. In July 2024, CMS officials initially estimated that the first year of the Demonstration would cost $5.2 billion. However, CMS officials told us in August 2025 that the first year of the Demonstration (2025) would cost an estimated $6.2 billion, the majority of which they expected to use to stabilize beneficiary premiums. Specifically, 97 percent (approximately $6 billion) was expected to subsidize premium stabilization, and the remaining 3 percent (approximately $200 million) was expected to support the additional financial protection for plan sponsors through the changes to the risk corridors. For the approximately $6 billion in estimated costs of Demonstration-related premium subsidies, we found that 33 of the 39 participating plan sponsors received $15 or less per beneficiary per month, based on our analysis of CMS data. The remaining six plan sponsors received $16 to $44 per beneficiary because their premium increases from 2024 to 2025 were higher than other plan sponsors.
Future year adjustments to and estimated costs of the demonstration. CMS officials told us they adjusted the parameters for the second year of the Demonstration (2026) based on the annual bids submitted by plan sponsors.[43] Specifically, CMS announced in July 2025 that the agency is doing the following for 2026.
· Providing a smaller premium reduction by reducing premiums for all participating standalone plans—and therefore beneficiaries—by $10 for 2026, less than the $15 in 2025.
· Increasing the limit on premium increases to $50 from 2025 to 2026, up from $35 in 2025.
· Eliminating the Part D risk corridor changes, reverting to current law thresholds and share of losses.[44]
CMS officials told us they designed the Demonstration so that they could adjust the parameters from one year to the next to reflect market conditions with the goal of reducing premium stabilization over time to facilitate a return to regular market conditions. Further, according to CMS officials, additional premium stabilization allows plan sponsors more time to adjust to the changing conditions in the standalone drug plan market and stabilize their bids while protecting beneficiaries from large, short-term premium changes.
CMS officials estimate the second year of the Demonstration, 2026, will cost $3.6 billion, bringing the total estimated cost of the Demonstration for the first and second years to $9.8 billion. The costs of the three previous Section 402 demonstrations that focused on Part D ranged from $0 (budget neutral) to $907 million. (See fig. 4.) (See app. I for more information on prior Section 402 demonstrations and their total costs).
Figure 4: Cost of Part D Premium Stabilization Demonstration and Previous Selected Demonstrations Focused on Medicare Part D
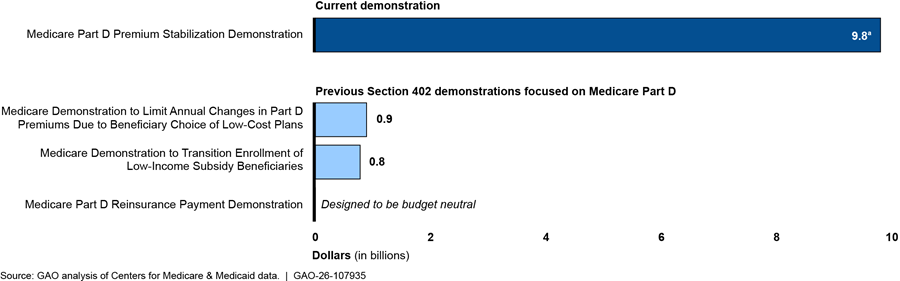
aThe approximate $9.8 billion cost of the Medicare Part D Premium Stabilization Demonstration covers the estimated costs of the first 2 years of the demonstration ($6.2 billion in 2025 and $3.6 billion in 2026). CMS has announced the demonstration will last at least 3 years, but the estimated costs of the third year (2027) are unknown as of September 2025. Cost estimates for previous demonstrations reflect 2025 dollars by adjusting for inflation based on the Congressional Budget Office’s projection of the Gross Domestic Product Index for 2025 and the actual Gross Domestic Product Index in prior years.
CMS’s Evaluation Is Being Designed to Determine Whether the Demonstration Achieved Its Goals While Accounting for Concurrent Part D Benefit Changes
The evaluation of the Part D Premium Stabilization Demonstration is being designed to determine whether the Demonstration achieved its goals of stabilizing beneficiary enrollment in standalone drug plans and stabilizing bids for standalone drug plans in future years, according to CMS officials. We calculated, based on CMS data, that the average monthly premium for standalone drug plan beneficiaries not eligible for the low-income subsidy increased from $42 in 2024 to $43 in 2025 under the Demonstration. Further, enrollment in standalone drug plans increased by approximately 2 percent from 2024 to 2025, and the percentage of all Part D enrollees in standalone drug plans remained at 42 percent. However, an evaluation is needed to determine the extent to which changes such as these to premiums and enrollment were due to the Demonstration and not to other factors.
Under an interagency agreement, CMS officials worked with ASPE officials to design a framework for evaluating the Demonstration, along with support from a contractor.[45] Designing the evaluation framework, or a starting point for evaluating the Demonstration, involves identifying potential data sources and methods for the evaluation. ASPE officials told us in September 2025 that, once the evaluation framework was finalized, a contractor will be tasked with implementing the evaluation. We received a copy of the finalized evaluation framework in January 2026. ASPE officials also told us they plan to continue their evaluation efforts in fiscal year 2026.
According to ASPE officials, they designed the evaluation to account for other factors that could have also affected the outcomes related to the Demonstration’s goals. These factors include, for example, concurrent changes to the Part D benefit that increased plan sponsors’ overall financial liability for Part D drug coverage in 2025 relative to 2024. CMS officials acknowledged these other factors and stated that the evaluation will work to distinguish the effects of the Demonstration from effects of other related changes.
ASPE officials told us that they worked with their contractor and sketched out broad strategies for building models from historic data of plan enrollments over time. Officials also said they will use these models to estimate what Part D plan enrollment patterns might have been had those other changes taken effect without the Demonstration. However, officials acknowledge that such models are necessarily based on assumptions, and it remains to be determined what specific models they will construct and how well those assumptions will be tested.
ASPE officials also shared that, because the Demonstration itself may change for each of the planned 3 years, any changes would need to be accounted for in the evaluation. For instance, in July 2025, CMS announced changes to the Demonstration’s parameters for its second year, 2026, as described above. To account for these annual changes, CMS officials told us they may need to evaluate the Demonstration on an annual basis, rather than across all years.
Because we received the finalized evaluation framework after we had concluded data collection for our study, we were unable to assess the quality of the evaluation framework. However, CMS’s plan to conduct an evaluation is consistent with the law authorizing the Demonstration. Specifically, Section 402 authorizes the Secretary to undertake demonstration projects to determine whether, and, if so, which of, the demonstration’s payment changes increase the efficiency and economy of Medicare services without reducing quality. Although the statute does not expressly require the agency to conduct an evaluation, an evaluation is necessary to determine the Demonstration’s effects. For instance, an evaluation would be necessary to more clearly discern the Demonstration’s effects on monthly premiums for and beneficiary enrollment in standalone drug plans in the years since the Demonstration began, in light of concurrent changes in the market. Further, the evaluation must be credibly designed such as to allow CMS to meaningfully measure the effects of the Demonstration compared to what would have occurred in its absence.
In addition, CMS officials told us they ultimately plan to use the findings from the evaluation of the Demonstration to inform future policy decisions related to Medicare Part D. The findings of the evaluation, and the evaluation process itself, may also help officials learn from the experience of this demonstration and apply such lessons learned to future Medicare demonstrations implemented under Section 402 authority.
Agency Comments
We provided a draft of this report to the Department of Health and Human Services for review and comment. The Department of Health and Human Services provided technical comments, which we incorporated as appropriate.
We are sending copies of this report to the appropriate congressional committees and the Secretary of Health and Human Services. In addition, the report is available at no charge on the GAO website at http://www.gao.gov.
If you or your staff have any questions about this report, please contact me at dickenj@gao.gov. Contact points for our Offices of Congressional Relations and Media Relations may be found on the last page of this report. Other major contributors to this report are listed in appendix II.

John E. Dicken
Director, Health Care
Appendix I: Examples of Medicare Demonstrations Implemented by the Centers for Medicare & Medicaid Services Under Section 402 Authority
The Secretary of Health and Human Services has broad authority to develop and implement demonstration projects to test certain changes to Medicare payment methods under section 402 of the Social Security Amendments of 1967 as amended (Section 402).[46]
The Centers for Medicare & Medicaid Services (CMS), the agency within the Department of Health and Human Services (HHS) responsible for overseeing the Medicare program, has conducted at least nine demonstrations using this authority to test policies that might address transitional issues associated with the implementation of major changes to the Medicare program, CMS officials told us. Four of these demonstrations sought, or are currently seeking, to test approaches to limit fluctuations in beneficiary premiums in Medicare Part D prescription drug benefit programs (see table 2). In addition, five other Section 402 demonstrations address other aspects of Medicare such as the Medicare Advantage Program (see table 3).
|
Demonstration name |
Time period |
Description |
Scope and participants |
Estimated budgetary effect (in 2025 dollars) |
|
Medicare Part D Premium Stabilization Demonstration |
2025 – TBD; Ongoing |
Designed to test whether stabilizing beneficiary premiums would stabilize beneficiary enrollment in standalone Part D prescription drug plans; and whether providing additional financial protection for plan sponsors during the implementation of provisions under the Inflation Reduction Act would help stabilize bids in future years. |
Voluntary nationwide demonstration open to all standalone Part D prescription drug plans. Announced July 29, 2024, with an application deadline of August 5, 2024. 782 out of 818 Part D prescription drug plans opted to participate in the demonstration in calendar year 2025.a |
(Estimated) $9.8 billionb |
|
Medicare Demonstration to Limit Annual Changes in Part D Premiums Due to Beneficiary Choice of Low-Cost Plans |
2007-2008 |
Designed to assess whether sudden changes in beneficiary premiums would affect beneficiary choices and participation in Medicare Advantage or Part D prescription drug plans, and whether maintaining stable beneficiary premiums results in Part D Sponsors bidding more accurately. |
Mandatory nationwide demonstration open to all Medicare Advantage and Part D prescription drug plans. |
(Estimated) $907 million |
|
Medicare Demonstration to Transition Enrollment of Low-Income Subsidy Beneficiaries |
2007-2008 |
Designed to test different approaches to calculate the benchmark that establishes low-income premium amounts and low-income subsidies and assess whether low-income Medicare beneficiaries would be able to maintain access to necessary prescription drug coverage. |
Mandatory nationwide demonstration open to all Medicare Advantage and Part D prescription drug plans. |
(Estimated) $795 million |
|
Medicare Part D Reinsurance Payment Demonstration |
2006-2010 |
Designed to test an alternative payment approach for coverage of prescription drugs, and incentivized plans to provide supplemental prescription drug coverage to Medicare beneficiaries. |
Voluntary nationwide demonstration open to all Medicare Advantage and standalone Part D prescription drug plans. Announced February 25, 2005, with an application deadline of March 23, 2005. 523 plans (2006); 538 plans (2007)c |
Designed to be budget neutral |
Source: Centers for Medicare & Medicaid Services (CMS). | GAO‑26‑107935
Note: Cost estimates for previous demonstrations reflect 2025 dollars by adjusting for inflation based on the Congressional Budget Office’s 2025 projection of the Gross Domestic Product Index and the actual Gross Domestic Product Index in prior years.
aCMS estimates that 99 percent of beneficiaries in Part D prescription drug plans in 2025 are covered by plan sponsors participating in the demonstration, with the remaining 1 percent of beneficiaries enrolled in employer group waiver plans.
bThe approximate $9.8 billion cost of the Medicare Part D Premium Stabilization Demonstration covers the estimated costs of the first 2 years of the demonstration ($6.2 billion in 2025 and $3.6 billion in 2026).
cEvaluation report enrollment analysis only included data for 2006 and 2007.
Table 3: Selected Demonstrations with Broader Application Across Medicare Programs, as of March 2025
|
Demonstration name |
Time period |
Description |
Number of participants |
Estimated budgetary effect (in 2025 dollars) |
|
Over-the-Counter COVID-19 Test Demonstration |
2022-2023 |
Designed to test the efficacy of Medicare covering over-the-counter COVID-19 tests without cost-sharing during the COVID-19 public health emergency. |
Mandatory nationwide demonstration open to all Medicare Advantage plans. |
Approximately, $1.2 billion over the course of the demonstration for an estimated 101 million COVID-19 tests for an estimated 8 million Medicare beneficiaries.a |
|
Medicare Advantage (MA) Quality Bonus Payment Demonstration |
2012-2014 |
Designed to test alternative methods for calculating and awarding bonuses to Medicare Advantage plans based on performance across select measures, and whether bonuses extended to plans with 3 or more stars would lead to more rapid and larger year-to-year improvements in their quality scores. |
Mandatory nationwide demonstration open to all Medicare Advantage plans. |
Nearly $14.7 billion in additional demonstration-related payments to MA contracts between 2012 and 2014 |
|
Multi-Payer Advanced Primary Care Practice Demonstration |
2011-2016 |
Designed as a joint effort with states across multiple payers to assess the effect of advanced primary care practices in utilization and expenditures for beneficiaries and improvements in quality and coordination of health care services received in advanced primary care practices. |
Offered in eight states (Maine, Michigan, Minnesota, New York, North Carolina, Pennsylvania, Rhode Island, and Vermont) More than 6,000 providers at more than 800 practices; beneficiaries. |
Designed to be budget neutral. |
|
Nursing Home Value Based Purchasing Demonstration |
2009-2012 |
Designed to test a value-based purchasing concept. Nursing home performance was assessed using measures from four domains: nurse staffing, quality outcomes, survey deficiencies, and potentially avoidable hospitalization rates. |
Offered in three states (Arizona, New York and Wisconsin) Approximately 182 nursing homes were selected to participate in the demonstration in the first year. |
Designed to be budget neutral. |
|
Medicare Preferred Provider Organization Demonstration |
2003-2005 |
Designed to test the impact of enhanced payment and risk-sharing arrangements between CMS and plans on the range of options and benefits available to beneficiaries. |
Offered in 21 states (Alabama, Arizona, Florida, Illinois, Indiana, Kansas, Kentucky, Louisiana, Maryland, Missouri, Nevada, New Jersey, New York, North Carolina, Ohio, Oregon, Pennsylvania, Rhode Island, Tennessee, Washington, West Virginia) 35 Preferred Provider Organization plans (selected by technical panel) |
Not included in the evaluation. |
Source: Centers for Medicare & Medicaid Services (CMS). | GAO‑26‑107935
Note: Cost estimates for these demonstrations reflect 2025 dollars by adjusting for inflation based on the Congressional Budget Office’s 2025 projection of the Gross Domestic Product Index and the actual Gross Domestic Product Index in prior years.
aData reflects estimates provided in 2023, the most recent cost estimate available.
John E. Dicken, dickenj@gao.gov
Staff Acknowledgments: In addition to the contact named above, William Black (Assistant Director), Kaitlin Asaly (Analyst-in-Charge), Patricia Broadbent, and Kimberly Perrault made key contributions to this report. Also contributing were Ariel Landa-Seiersen, Laurie Pachter, Eric Peterson, Jennifer Rudisill, Ravi Sharma, Roxanna Sun, and Jeffrey Tamburello.
The Government Accountability Office, the audit, evaluation, and investigative arm of Congress, exists to support Congress in meeting its constitutional responsibilities and to help improve the performance and accountability of the federal government for the American people. GAO examines the use of public funds; evaluates federal programs and policies; and provides analyses, recommendations, and other assistance to help Congress make informed oversight, policy, and funding decisions. GAO’s commitment to good government is reflected in its core values of accountability, integrity, and reliability.
Obtaining Copies of GAO Reports and Testimony
The fastest and easiest way to obtain copies of GAO documents at no cost is through our website. Each weekday afternoon, GAO posts on its website newly released reports, testimony, and correspondence. You can also subscribe to GAO’s email updates to receive notification of newly posted products.
Order by Phone
The price of each GAO publication reflects GAO’s actual cost of production and distribution and depends on the number of pages in the publication and whether the publication is printed in color or black and white. Pricing and ordering information is posted on GAO’s website, https://www.gao.gov/ordering.htm.
Place orders by calling (202) 512-6000, toll free (866) 801-7077,
or
TDD (202) 512-2537.
Orders may be paid for using American Express, Discover Card, MasterCard, Visa, check, or money order. Call for additional information.
Connect with GAO
Connect with GAO on X,
LinkedIn, Instagram, and YouTube.
Subscribe to our Email Updates. Listen to our Podcasts.
Visit GAO on the web at https://www.gao.gov.
To Report Fraud, Waste, and Abuse in Federal Programs
Contact FraudNet:
Website: https://www.gao.gov/about/what-gao-does/fraudnet
Automated answering system: (800) 424-5454
Media Relations
Sarah Kaczmarek, Managing Director, Media@gao.gov
Congressional Relations
David A. Powner, Acting Managing Director, CongRel@gao.gov
General Inquiries
[1]See Pub. L. No. 117-169, tit. I, subtit. B, pt. 3, § 11201(d), 136 Stat. 1818, 1888 (codified as amended at 42 U.S.C. § 1395w-113(a)(8)).
[2]The expected 179 percent increase in costs included both standalone drug plans and MA drug plans.
[3]The IRA’s 6 percent cap applies only to the base beneficiary premium, which is calculated as a percentage of the nationwide average of Part D plans’ expected costs of providing Part D benefits. Part D drug plans with bids that exceed the nationwide average may charge an additional premium.
[4]Section 402(a)(1)(A) of the Social Security Amendments of 1967 as amended (Section 402), authorizes the Secretary of Health and Human Services to carry out demonstration projects “to determine whether, and if so, which changes in methods of payment or reimbursement” under Medicare “would have the effect of increasing the efficiency and economy of health services” covered under Medicare “through the creation of additional incentives to these ends without adversely affecting the quality of such services.” See Pub. L. No. 90-248, § 402, 81 Stat. 821, 930 (1968), as amended by the Social Security Amendments of 1972, Pub. L. No. 92-603, § 222, 86 Stat. 1329, 1390 (codified at 42 U.S.C. § 1395b-1) (hereinafter Section 402). Section 402 demonstration authority expressly applies to Part D. See 42 U.S.C. § 1395w-152.
[5]Specifically, we found that the Demonstration, as implemented in 2025, created additional incentives for participating Part D plan sponsors to increase the economy and efficiency of Medicare services and enabled the agency to determine whether those changes in payment methods increased the efficiency and economy of Medicare services without adversely affecting quality, consistent with the requirements of Section 402. GAO, Department of Health and Human Services—Medicare Part D Premium Stabilization Demonstration, B-336645 (Washington, D.C.: May 27, 2025).
[6]We focused our analyses on beneficiaries enrolled in standalone drug plans that existed in both 2024 and 2025 (including beneficiaries in plans in 2024 who would have been transferred to another plan in 2025) when looking at how premiums would change absent the Demonstration if beneficiaries had remained in their plan in 2025. We excluded enrollees of employer group waiver plans, which do not submit the bid data used to calculate premiums. We focused our analyses on beneficiaries who did not receive the Part D low-income subsidy because non-low-income subsidy beneficiaries generally are required to pay Part D premiums and therefore are directly affected by annual premium increases; this included approximately 80 percent of beneficiaries enrolled in standalone plans in 2024 and 2025. Beneficiaries eligible for the low-income subsidy include those whose income is below 150 percent of the federal poverty level or who receive Supplemental Security Income benefits and can enroll in certain Part D plans without paying a premium. Employer group waiver plans are a type of MA plan designed for employers and unions to offer customized Medicare benefits to their retirees.
[7]When calculating average premiums in 2024 and 2025 for beneficiaries who were not eligible for the low-income subsidy, we weighted our results by enrollment of these beneficiaries in each year. When calculating how premiums would change from 2024 to 2025 for beneficiaries not eligible for the low-income subsidy who remained in their plan or were transferred to another one, we weighted our results by 2024 enrollment.
[8]We identified and interviewed plan sponsors that had the largest number of Part D beneficiaries enrolled in standalone drug plans in 2024; collectively, these plan sponsors operated standalone drug plans that served more than 18 million beneficiaries (79 percent) of the 22.9 million beneficiaries enrolled in standalone drug plans in July 2024.
[9]Within each PDP region, plan sponsors can provide prescription drug coverage, but cost and coverage options can vary.
[10]Whether a beneficiary chooses traditional Medicare or MA depends on a variety of factors, including the Part D premium. For example, some beneficiaries may choose to enroll in traditional Medicare because they want greater choice to select the doctor or hospital of their choosing, as long as the doctor or hospital accepts Medicare. Beneficiaries enrolled in MA plans may be restricted to using doctors in the plan’s network and may need referrals for specialists. In addition, some beneficiaries may choose to enroll in MA plans to obtain benefits that MA plans may offer, such as vision, hearing, and dental services that are not available in traditional Medicare.
[11]All plan sponsors must cover a wide range of drugs commonly prescribed to Medicare beneficiaries. A plan’s covered drug list includes brand-name drugs—those marketed under exclusive, trademark-protected names and patents. Plan sponsors also cover generic versions of brand-name drugs, which may use the same active ingredients as brand-name drugs but are marketed without the brand-name on the label and may be sold at a lower cost than the brand-name drug. The amount a beneficiary pays varies by the coverage phase and whether the drug is a brand-name or generic.
[12]Under the standard benefit, the beneficiary is responsible for paying a monthly premium, cost sharing in the form of an annual deductible, and copayments or coinsurance for drug purchases. A deductible is a fixed dollar amount that beneficiaries must pay before coverage takes effect. Beneficiaries with supplemental drug coverage or who are eligible for the low-income subsidy may not pay the full deductible amount. Additionally, some drug plans do not have a deductible.
[13]This monthly prospective payment is increased or decreased depending on whether the beneficiary’s clinical diagnoses indicate that their drug costs are likely to be higher or lower than the average beneficiary—a process called risk adjustment.
[14]Part D plan sponsor bids reflect the expected costs of providing the Part D benefit to a beneficiary with average prescription drug costs. In general, the beneficiary premium for a Part D plan equals the sum of the base beneficiary premium and the difference between the plan’s bid amount and the nationwide average bid amount.
The national average bid amount calculation does not include Part D bids submitted by certain types of plans, such as those for beneficiaries with specialized needs (called “special needs plans”), Medicare Medical Savings Account plans, and MA private fee-for-service plans.
[15]Under section 1860D-13 of the Social Security Act, as added by section 11201 of the IRA, the base beneficiary premium for 2024 through 2029 is equal to the lesser of the prior year’s base beneficiary premium increased by 6 percent, or the base beneficiary premium as it would have been calculated if the IRA’s premium stabilization provision had not been enacted. See 42 U.S.C. § 1395w-113(a)(8). The Medicare program—through additional subsidies to Part D plan sponsors—covers the cost of the 6 percent annual increases in the base beneficiary premium.
[16]See Madeleine Cline and David M. Liner, Navigating New Waters: How the Inflation Reduction Act Alters Government Funding for Medicare Part D. (Seattle, Wash.: Milliman, Inc., Aug. 6, 2024).
[17]Beneficiaries eligible for the low-income subsidy (e.g., those who receive Supplemental Security Income benefits or who meet residency and enrollment requirements whose income is below 150 percent of the federal poverty level) can enroll in certain plans, known as benchmark plans, without paying a premium.
[18]Pub. L. No. 117-169, tit. I, subtit. B, 136 Stat. 1818, 1833-1905.
[19]Out-of-pocket costs vary based on utilization of prescription drugs. For example, ASPE reported that annual out-of-pocket costs were highest for beneficiaries with certain health conditions, such as cystic fibrosis ($7,800), followed by multiple myeloma and other cancers ($5,700). See Office of the Assistant Secretary for Planning and Evaluation, Inflation Reduction Act Research Series—Projecting the Impact of the $2,000 Part D Out-Of-Pocket Cap for Medicare Part D Enrollees with High Prescription Drug Spending, Issue Brief. (Washington, D.C.: Jan. 2025). Under the IRA, beginning in 2025, beneficiaries have the option to pay their out-of-pocket costs in monthly installments over the course of the calendar year, through the Medicare Prescription Payment Plan.
[20]42 U.S.C. § 1395b-1(a)(1)(A).
[21]CMS officials told us these nine demonstrations were representative of the budgetary impact, size, and scope of Section 402 demonstrations that CMS has conducted since 1995 but may not be exhaustive, in part because of (1) the fact that demonstrations have been conducted by various CMS components over that time period and (2) changing record-keeping practices within the requested time period.
[22]All standalone plans, including employer group waiver plans could opt to participate in the demonstration. Because employer group waiver plans do not submit bids and are not subject to risk corridor reconciliation, they are not eligible for the year-over-year plan premium limit or risk corridor elements of the demonstration.
[23]These results are weighted based on 2024 beneficiary enrollment in standalone drug plans.
[24]In 2024, plan sponsors received $20.2 billion in direct subsidy payments and $68.3 billion in reinsurance payments; comparatively, in 2025, plan sponsors are estimated to receive $104.0 billion in direct subsidy payments and $29.0 billion in reinsurance payments. See 2025 Annual Report of the Boards of Trustees of the Federal Hospital Insurance Trust Fund and the Federal Supplementary Medical Insurance Trust Fund (June 18, 2025: Washington, D.C.).
[25]See Medicare Payment Advisory Commission, The Medicare Prescription Drug Program (Part D): Status Report (Ch. 12), March 2025.
[26]For example, beneficiaries have increasingly used the antidiabetic class of glucagon-like peptide-1 (GLP-1) agonists in recent years.
[27]See Cline and Liner, Navigating New Waters.
[28]CMS officials noticed greater-than-expected variation among the standalone drug plan bids, relative to the MA drug plan bids.
[29]These calculations are weighted using July 2024 and July 2025 enrollment data for non-low-income subsidy beneficiaries and exclude beneficiaries of employer group waiver plans because these plans do not submit bid data.
[30]For plans with enhanced benefits beyond the standard benefit package, the premium also reflects the additional expected cost of these benefits.
[31]See Medicare Payment Advisory Commission, Report to the Congress: Medicare and the Health Care Delivery System, June 2025.
[32]As previously noted, premiums for standalone drug plan beneficiaries in 2024 who remained in their plan would have increased from $43 in 2024 to $81 in 2025. In contrast, the average monthly Part D premium for MA drug plans were significantly lower. For example, average monthly MA drug plan premiums were $9 in 2024 and $7 in 2025. These estimates were weighted by enrollment, exclude enrollees in employer group plans and plans for beneficiaries with certain health care needs (“special needs plans”), and reflect premium reductions implemented by MA plans during the MA bidding process. See Juliette Cubanski and Anthony Damico, Key Facts About Medicare Part D Enrollment, Premiums, and Cost Sharing in 2025, (San Francisco, Calif.: KFF, July 16, 2025).
[33]CMS officials cited a previous study, which found that 28 percent of Part D enrollees in plans with premium increases of at least $20 switched plans in a given year between 2006 and 2010, compared to 7 to 8 percent of enrollees who faced a premium increase of $10 or less or a premium reduction. See Jack Hoadley et al. “To Switch or Not to Switch: Are Medicare Beneficiaries Switching Drug Plans to Save Money?” KFF Issue Brief (San Francisco, Calif.: KFF, Oct. 10, 2013).
[34]Plan sponsors’ enrollment projections for the upcoming year are subject to uncertainty and therefore may differ from actual enrollment in that year. For example, Part D bids for 2024 projected an 18 percent decline in standalone plan enrollment from 2023 to 2024, and actual enrollment declined by about 2 percent.
[35]If an MA plan’s estimated costs for Medicare-covered services are below the maximum amount CMS will pay private plans in an area (known as the benchmark), the MA plan retains a portion of the difference, known as a “rebate.” MA plan sponsors can use the rebate dollars from these payments to lower their Part D premiums for beneficiaries. See Nancy Ochieng, Meredith Freed, Jeannie Fuglesten Biniek, Anthony Damico, and Tricia Neuman, “Medicare Advantage in 2025: Premiums, Out-of-Pocket Limits, Supplemental Benefits, and Prior Authorization,” (San Francisco, Calif.: KFF, July 28, 2025).
[36]CMS officials also said that the anticipated monthly premium increases resulting in widespread enrollment changes could result in administrative, operational, and financial burdens for the agency. For instance, officials said processing substantial enrollment changes is resource-intensive for CMS, requiring system adjustments, increased customer service demands, and heightened oversight.
[37]Plan sponsors can change the formularies in their drug plans each year, so it is also possible that even if beneficiaries remain in the same standalone drug plan each year, they may not have access to the same medications from year to year.
[38]Because MA drug plans have more tools, such as rebates, to manage Part D costs, CMS officials told us they limited the demonstration to standalone drug plans.
[39]If the $15 reduction resulted in a standalone drug plan’s premium being below $0, the plan’s Part D premium was reduced to the point where the plan’s total premium equaled $0. Further, the $15 reduction applied to all standalone drug plans that opted into the demonstration, including employer group waiver plans.
[40]The $35 limit does not apply to employer group waiver plans, in part because the plans do not submit to CMS bids that the agency uses to calculate premiums.
[41]These average monthly premiums were weighted by beneficiary enrollment in 2024 and 2025, respectively.
We also found that the average monthly premium (weighted by 2024 enrollment) for standalone drug plan beneficiaries not eligible for the low-income subsidy who remained in their same plan from 2024 to 2025 would have increased from $43 in 2024 to $49 in 2025. The $49 average premium in 2025 is lower than the average monthly premium of $81 that would have occurred absent the Demonstration for these beneficiaries if they had remained in the same plan.
[42]CMS officials also stated that the additional financial protection for plan sponsors provided a financial incentive for plan sponsors to participate in the Demonstration despite potentially enrolling beneficiaries with higher drug costs than expected.
[43]CMS officials told us that announcing the Demonstration parameters after receiving the bids for the upcoming year helps promote a competitive bidding environment. This is because announcing the Demonstration parameters in advance of bid submissions could, for example, incentivize plans sponsors to raise premiums above what they would otherwise to maximize their Demonstration-related subsidy.
[44]Centers for Medicare & Medicaid Services, Annual Release of Part D National Average Bid Amount and Other Part C & D Bid Information (July 28, 2025).
[45]ASPE is responsible for advising HHS on health policy development and providing advice and analysis on economic policy. Among other things, the office coordinates HHS’s evaluation, research, and demonstration activities by conducting research and evaluation studies, developing policy analyses, and estimating the cost and benefits of policy alternatives under consideration by HHS. HHS announced in March 2025 that it will merge ASPE with the Agency for Healthcare Research and Quality to create the Office of Strategy, although the department had not fully implemented the reorganization as of January 2026.
ASPE contracted with a contractor to help develop the evaluation design.
[46]See 42 U.S.C. § 1395b-1(a)(1)(A).
